비용을 동반한 만성비부비동염 환자에서 Calcitonin Gene-Related Peptide의 발현 및 역할
Expression and Role of Calcitonin Gene-Related Peptide in Patients With Chronic Rhinosinusitis With Nasal Polyposis
Article information
This article has been corrected. See "Expression and Role of Calcitonin Gene-Related Peptide in Patients With Chronic Rhinosinusitis With Nasal Polyposis" on page 190.
Trans Abstract
Background and Objectives
A recent study revealed that calcitonin gene-related protein (CGRP) plays an important role in inflammatory airway diseases. However, the influence of CGRP on chronic rhinosinusitis (CRS) has not been studied. This study investigated the expression, activity, and potential pathogenic role of CGRP in patients with CRS with nasal polyposis (CRSwNP).
Subjects and Method
Patients with CRSwNP and control subjects were enrolled. The CRSwNP group was divided according to the presence of eosinophilic polyps and non-eosinophilic polyps. Nasal polyps (NPs) and uncinate tissues (UTs) from patients with CRSwNP and UTs from control subjects were obtained to investigate the expression of α-/β-CGRP and chromogranin A. In addition, the expression patterns of cytokines following exposure to exogenous CGRP were analyzed in dispersed nasal polyp cells (DNPCs) from patients with eosinophilic or non-eosinophilic CRSwNP. The effects of CGRP on lipopolysaccharide (LPS)-induced nuclear factor-kappa light chain enhancer of activated B cells (NF-κB) signaling change were evaluated in THP-1 cells.
Results
The expression of α-/β-CGRP and number of CGRP-producing cells were significantly higher in NPs from patients with CRSwNP than in UTs from controls. Exogenous CGRP decreased the expression of inflammatory cytokines and increased that of the anti-inflammatory cytokines in DNPCs from patients with eosinophilic nasal polyps (EPs) and also increased the expression of tissue remodeling-related and anti-inflammatory cytokines in DNPCs from patients with non-eosinophilic nasal polyps (N-EPs). CGRP inhibited the nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor (IκB) phosphorylation and NF-κB translocation in LPS-stimulated M1 macrophages.
Conclusion
CGRP expression in NPs may play a significant role in nasal polypogenesis through inflammatory modulation, and it could be a future target to modulate certain aspects of CRSwNP.
Introduction
Chronic rhinosinusitis (CRS) is a persistent inflammatory disease of nasal and paranasal sinus mucosa [1]. CRS can be classified phenotypically based on the presence of nasal polyps (NPs) into CRS without NP (CRSsNP) or CRS with NP (CRSwNP), the latter of which is currently classified into two major endotypes: eosinophilic and non-eosinophilic CRSwNP. These types have different relationships with the pathogenesis of CRS, inflammatory cell and T-cell patterns, tissue remodeling, and epithelial barrier function [2]. Neuropeptides are small proteins produced by neurons and immune cells that act on G protein-coupled receptors [3]. These neuropeptides influence the regulation of immune responses by promoting chemokine expression, and they are also involved in the suppression of immune responses by inhibiting pro-inflammatory and inflammatory cytokines and transcription factors in the airway [4,5]. Neuropeptides released from sensory and parasympathetic nerves of the nasal mucosa are considered important in the development of polyposis [6]. For instance, capsaicin, which acts on C fibers, was applied topically for the possible prevention of NP recurrence, and the results indicated that capsaicin treatment improved nasal symptoms and reduced polyp size compared to the effects of control [7].
Among these neuropeptides, calcitonin gene-related protein (CGRP) is a 37-amino acid neuropeptide, and two isoforms of CGRP, α- and β-CGRP are known. It has been reported α-CGRP is mainly expressed in central and peripheral nervous systems and neuroendocrine cells, β-CGRP is expressed in immune cells. Although the site of expression is different, the biological activity are known to be the same [8]. A recent study reported that CGRP and chromogranin A (CgA) were expressed in pulmonary neuroendocrine cells, which stimulates type 2 innate lymphoid cells and Th2 cells to increase interleukin (IL)-5 expression, inducing type 2 inflammation [9]. In addition, IL-33–induced airway inflammation is negatively regulated by treatment with CGRP [10]. It is known that CGRP regulates macrophages polarization, which is involved in the inhibition of M1 macrophage activity and promotion of M2 macrophage (alternative macrophage) activity [11,12]. CGRP also inhibits the activity of nuclear factor-kappa light chain enhancer of activated B cells (NF-κB) through protein kinase A pathways in M1 macrophages, reducing the expression of pro-inflammatory cytokines [12]. Recent research demonstrated that CGRP also inhibited the expression of nucleotide binding oligomerization domain-like receptor pyrin domain-containing protein 3, IL-1β, and macrophage inflammatory protein-1 in BALB/c mouse corneas and macrophages infected by Aspergillus fumigatus [13]. However, no study examined the expression or immunologic roles of CGRP in the pathogenesis of CRSwNP despite the large number of studies focused on lower respiratory tract diseases.
This study investigated whether the expression of CGRP is elevated in NPs and examined the potential role of CGRP in the pathogenesis of CRSwNP.
Subjects and Methods
Patients
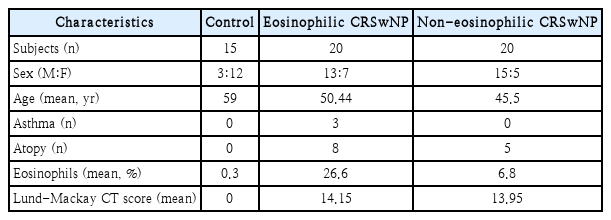
A total of 55 patients were recruited for this study. The number of patients with eosinophilic CRSwNP, non-eosinophilic CRSwNP, and control subjects were 20, 20, and 15 respectively. As a control group, patients who underwent other endoscopic surgeries, such as dacryocystectomy, endoscopic skull base surgery, or endoscopic orbital surgery, were recruited.
CRSwNP was diagnosed according to the European Position Paper on Rhinosinusitis and NPs 2012 guidelines [1]. NPs were divided into eosinophilic NPs (EPs) and non-eosinophilic NPs (N-EPs). In hematoxylin and eosin staining, NPs were classified as EPs if eosinophils comprised over 10% of the total inflammatory cell population per high-power field (HPF), and the others were classified as N-EPs. Diagnosis of the atopic status was conducted by the multiple allergen simultaneous tests or skin prick test. Bronchial asthma was diagnosed by pulmonologist consultation.
Patients who took a medication (e.g., anti-leukotrienes, oral steroid, antibiotics) within 4 weeks before surgery, caught a upper respiratory tract infections, and underwent revision endoscopic sinus surgery were excluded. The clinical characteristics of patients were described in Table 1. All patients provided written informed consent prior to study enrollment. The study was approved by the Institutional Review Board of Chungnam National University Hospital (IRB File No: 20-12-078).
THP-1 cell culture and activation
Human monocytes, THP-1 cells were cultured in RPMI 1640 medium (GenDEPOT, Barker, TX, USA) which was supplemented with 10% heat inactivated fetal bovine serum (FBS) (Gibco Laboratories, Grand Island, NY, USA), streptomycin (100 μg/mL; Sigma-Aldrich, St. Louis, MO, USA), and penicillin (100 IU/mL; Sigma-Aldrich). THP-1 cells are differentiated into macrophages via 48 hours of incubation with 200 nM phorbol 12-myristate 13-acetate (PMA) followed by 24 hours of incubation in serum-free RPMI 1640 medium. After starvation, cells were treated with 100 nM CGRP for 30 minutes followed by incubation with 1 μg/mL lipopolysaccharide (LPS) for 24 hours.
IL-1β and IL-6 levels in THP-1 cell supernatant were measured according to the manufacturer’s instructions using human IL-1β (R&D Systems, Minneapolis, MN, USA) and IL-6 ELISA kit (Abcam, Cambridge, UK).
Tissue preparation
Uncinate tissues (UTs) were obtained from all participants. Tissues of NP were collected from the patients with CRSwNP. Some portion of the NP and UT samples from each subject were fixed with 4% paraformaldehyde, embedded in paraffin, and sectioned to 4 μm thickness. Tissue sections were mounted onto saline-coated micro slides (Muto Pure Chemicals, Tokyo, Japan), and used for immunofluorescence (IF) test and immunohistochemistry (IHC) analysis subsequently. In addition, the remaining NP and UT samples were immediately soaked in 1× RIPA buffer (Cell Signaling Technology, Danvers, MA, USA) containing protease inhibitor (Cell Signaling Technology) for downstream protein extraction. Two fresh NPs were obtained from one eosinophilic CRS sample and one non-eosinophilic CRS sample and used to culture dispersed nasal polyp cells (DNPCs).
IHC staining
Tissue sections were deparaffinized and rehydrated with graded alcohol series. Endogenous peroxidase activity was blocked with 3% H2O2 for 15 min, and samples were washed with phosphate-buffered saline (PBS). By boiling the slides in 10 mM citrate buffer (pH 6.0) for 5 min, antigen retrieval was performed. Slides were incubated with anti–α-CGRP (mouse, 1:200; ROCKLAND, Limerick, PA, USA) and anti–β-CGRP (rabbit, 1:200; MyBioSource, San Diego, CA, USA) as primary antibodies at 4°C overnight. PBS was used instead of primary antibodies in the negative controls. The slides were incubated with anti-mouse HRP-conjugated (1:300; Santa Cruz Biotechnology, Dallas, TX, USA) and anti-rabbit HRP-conjugated (1:300; Santa Cruz Biotechnology) secondary antibody for 1 hour at room temperature. Sections were developed with 3, 3′-diaminobenzidine solution (DAKO, Brüsseler, Germany) for 2 min at room temperature. We examine the slides under an optical microscope (Olympus, Tokyo, Japan). α-/β-CGRP–positive cells were counted at five different sites in each tissue under HPF (×400).
IF staining
Sections were incubated for 1 h in streptavidin-biotin blocking solution (Vector Laboratories, Burlingame, CA, USA) and then stained with anti–α-CGRP (rabbit, 1:200; antibodies. com, Cambridge, UK) and anti-CgA (mouse, 1:200; LSBio, Seattle, WA, USA). Negative controls were prepared using PBS in place of the primary antibodies. Alexa Fluor 488-conjugated anti-mouse IgG (1:300; Invitrogen, Carlsbad, CA, USA) and Alexa Fluor 594-conjugated anti-rabbit IgG (1:300; Invitrogen) were then used as secondary antibodies. For nuclear counterstaining, 4′, 6-Diamidino-2-phenylindole (Invitrogen) was applied at a concentration of 300 nM. We mounted the specimens in VectaShield Antifade Mounting Medium (CH-1000; Vector Laboratories) and observed the slides under a fluorescence microscope (Olympus). Cells positive for both α-CGRP and CgA were counted per HPF (×1000) at five different sites in the tissue.
Western blotting
For lysing the tissues and THP-1 cells, we use Pro-Prep solution (iNtRON, Seongnam, Korea). Total protein was measured by using the Bradford protein assay (Bio-Rad Laboratories, Hercules, CA, USA). Electrophoresis was performed to separate proteins using 15% sodium dodecyl sulfate-polyacrylamide gel. Proteins were transferred onto polyvinylidene difluoride membranes (Immobilon-P; Millipore, Burlington, MA, USA). After blocking with 5% skim milk at room temperature for 30 min, the membrane was incubated overnight at 4°C with specific antibodies against α-CGRP (rabbit, 1:2000; antibodies.com), β-CGRP (rabbit, 1:200; MyBioSource), CgA (mouse, 1:2000; LSBio), NF-κB (rabbit, 1:1000; Cell Signaling Technology), nuclear factor of kappa light polypeptide gene enhancer in B cells inhibitor (IκB) (mouse, 1:1000; Cell Signaling Technology), tubulin (rabbit, 1:3000; Cell Signaling Technology), lamin B1 (rabbit, 1:3000; Abcam) and glyceraldehyde-3-phosphate dehydrogenase (rabbit, 1:3000; Cell Signaling Technology). After three times washing, incubating the membranes for 1 h at room temperature was performed, with the corresponding secondary antibodies (goat anti-mouse/rabbit, 1:3000; Cell Signaling Technology). The Chemiluminescence with Amersham ECL plus western blotting detection system (GE Healthcare, Brooklyn, NY, USA) was used.
DNPCs culture
DNPCs were prepared from NPs as previously described [14]. A gentle MACSTM Digested tissue was dissociated into single-cell suspension using Dissociator (Miltenyi Biotec, Bergisch Gladbach, Germany). The suspensions were filtered using cell strainers with a pore size of 70 μm (SPL, Pocheon, Korea). After twice washing with culture medium (Dulbecco’s modified Eagle’s medium [Gibco Laboratories] containing 10% FBS, 100 IU/mL penicillin, 100 μg/mL streptomycin), the cell pellets were re-suspended in Red Blood Cell Lysis Buffer (Roche, Mannheim, Germany). After washing, cells were plated in 12-well culture plates (SPL) at a density of 3×106 per well. Cells were cultured in the absence or presence of 100 nM α-CGRP (Eurogentec, Seraing, Belgium). The cells and supernatants were harvested after 48 h for cytokine profile study.
Cytokine profile
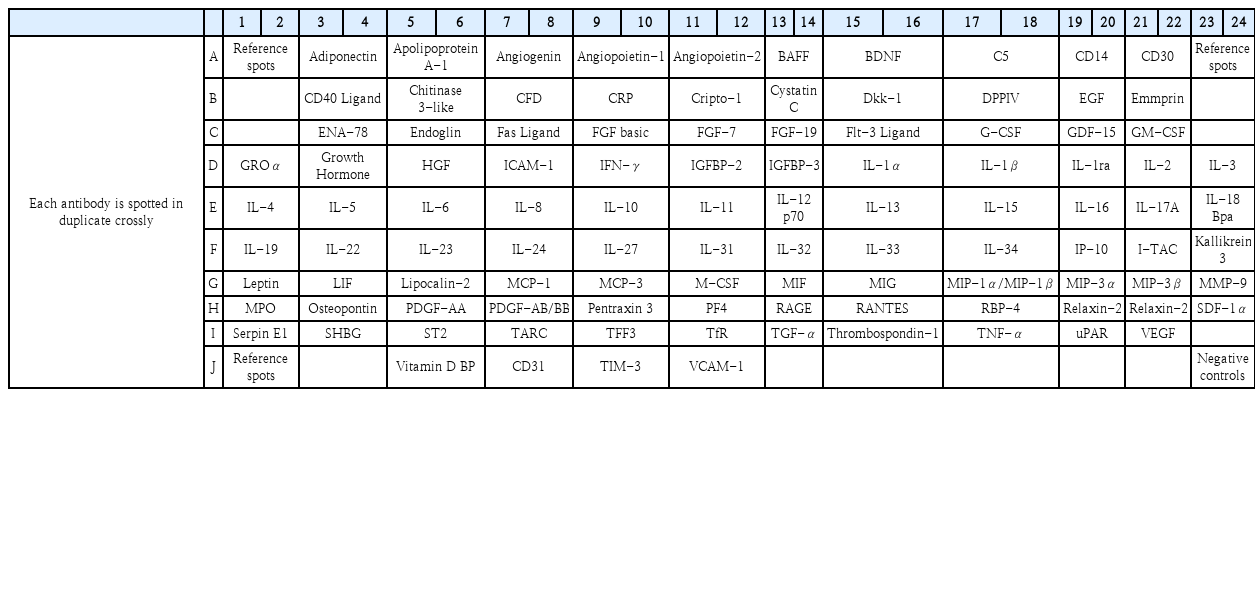
Cellular suspensions were obtained from prepared DNPCs. We examined the cytokines in the secretome by using the Proteome Profiler Assay Human XL Cytokine Array Kit (R&D Systems) according to manufacturer’s suggestion. The reference key for the cytokine array was adapted from the manufacturer’s information in Table 2. To determine the mean gray values of duplicate cytokine dots of gray-scale images, we used the HLImage++ for array analysis [15]. Since the patient’s tissue samples were insufficient, the experiment was conducted with one sample for each type.
Statistical analysis
All data were analyzed by using GraphPad Prism v8.00 (GraphPad Software Inc., San Diego, CA, USA), and student’s t-test. For the images, program Image J (National Institutes of Health, Bethesda, MD, USA) was used to adjust the sensitivity of the parts that were shown double positive, and then count the specific cells. The data are expressed as the mean±standard error of mean (SEM). Statistical significance was defined as p<0.05, and the degree of significance was presented as follows: *p<0.05; †p<0.01; ‡p<0.001; §p<0.0001.
Results
Expressions of α-/β-CGRP was increased in the NPs
IHC was conducted to examine α-/β-CGRP expression in tissues in the groups (Fig. 1A). The numbers of positive cells were counted separately in the epithelial and subepithelial layers, which is shown in Fig. 1B and C. The numbers of α-/β-CGRP–positive cells were significantly higher in the NP tissues in the CRSwNP group than in UTs from the control and CRSwNP groups (Fig. 1B and C). However, there were no significant differences in α-/β-CGRP expression in NPs between the ECRS and NECRS groups (Fig. 1B and C). α-/β-CGRP expression was also evaluated via Western blotting (Fig. 2A), and α-/β-CGRP expression was significantly higher in NPs in the CRSwNP group than in UTs in the control group, but there were no significant differences between the two endotypes of NPs (Fig. 2B and C). The correlations between α-/β-CGRP expression and the Lund-Mackay (L-M) CT scores of patients were analyzed. There were positive correlations between α-CGRP expression and the L-M CT score in both the ECRSwNP and NECRSwNP groups (Fig. 2D). However, there was no significant correlation between β-CGRP expression and the L-M CT score (Fig. 2E).
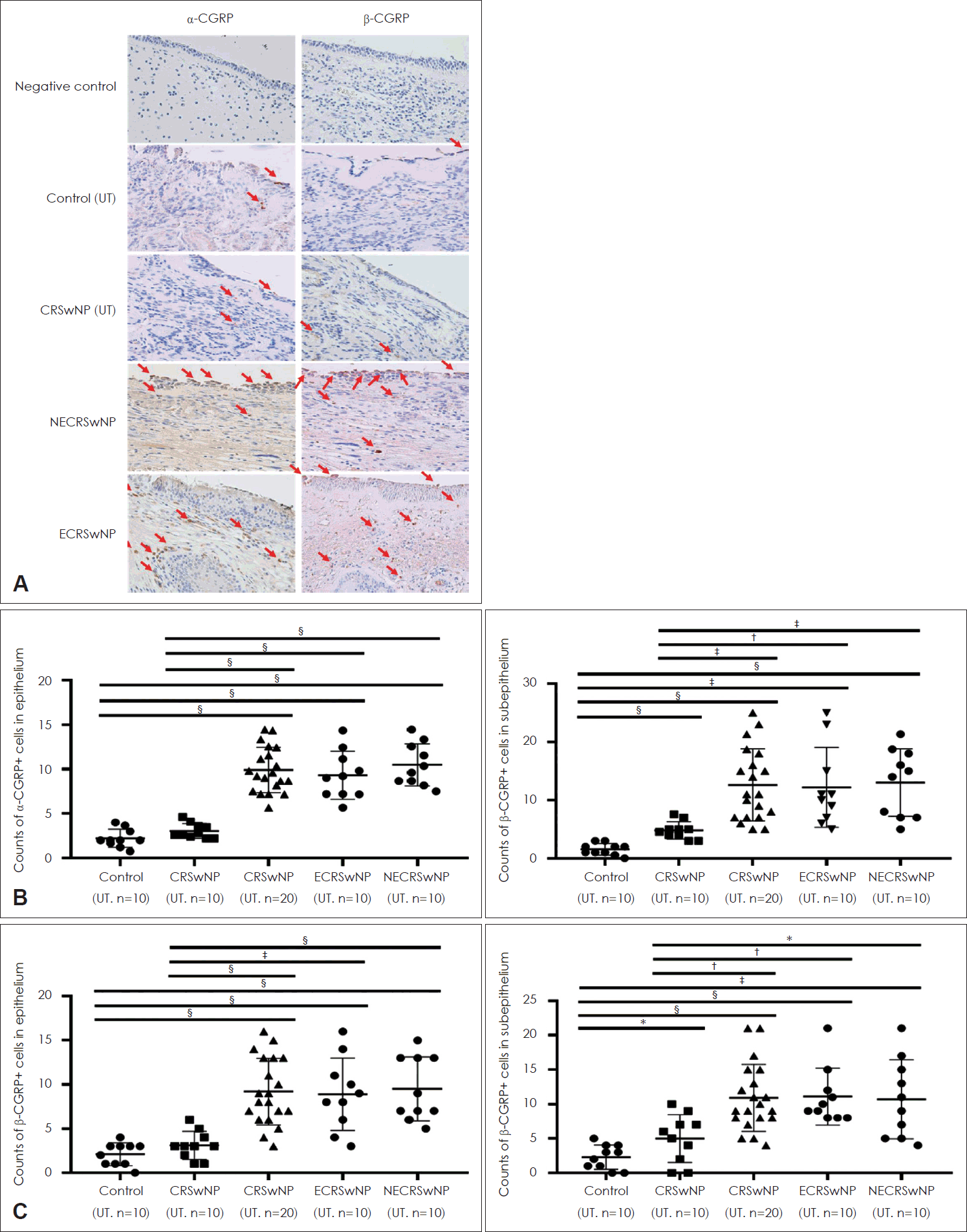
Expression of α-/β-CGRP among the different groups. A: Representative image of α-/β-CGRP expressions among groups using immunohistochemistry (original magnification, ×400). B and C: The number of α-/β-CGRP positive cells in the tissue among the different groups. *p<0.05; †p<0.01; ‡p<0.001; §p<0.0001. CGRP, calcitonin gene-related peptide; UT, uncinated tissue; CRSwNP, chronic rhinosinusitis with nasal polyp; NECRSwNP, non-eosinophilic CRSwNP; ECRSwNP, eosinophilic-CRSwNP.
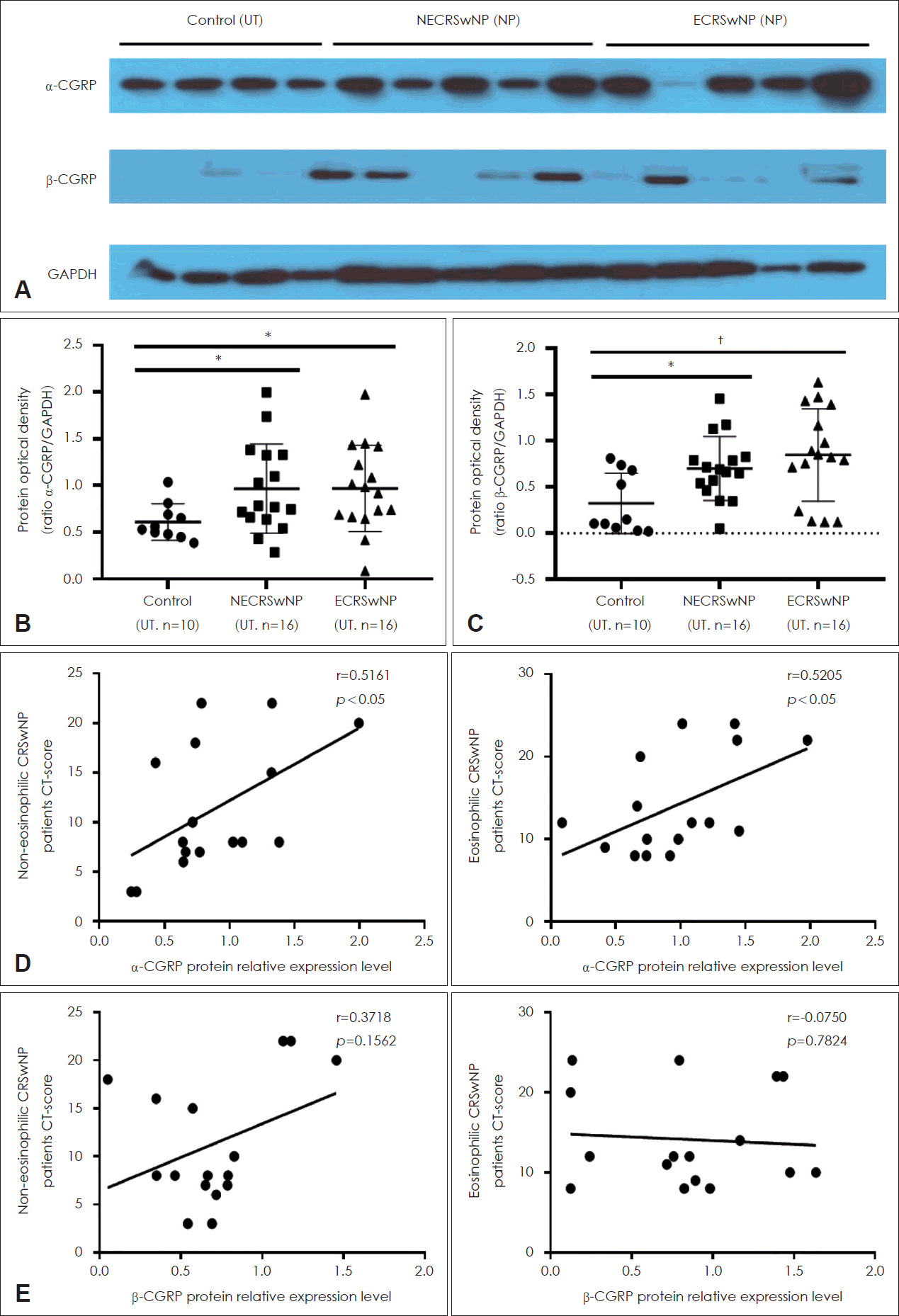
Expression of α-/β-CGRP in the tissues from uncinated tissues and nasal polyp tissues, and its correlation with L-M CT-score in the patients with CRSwNP. A-C: Representative image of α-/β-CGRP expressions using western blot assay in the tissues from UT and nasal polyp tissues. D: Correlation between L-M CT-score and α-CGRP in the patients with eosinophilic and non-eosinophilic CRSwNP. E: Correlation between L-M CT-score and β-CGRP in patients with eosinophilic and non-eosinophilic CRSwNP. *p<0.05; †p<0.01. UT, uncinated tissue; NECRSwNP, non-eosinophilic chronic rhinosinusitis with nasal polyp; ECRSwNP, eosinophilic CRSwNP; NP, nasal polyp; CGRP, calcitonin gene-related peptide; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; L-M, Lund-Mackay.
CGRP-producing neuroendocrine cell counts were higher in the CRSwNP group
The expression of CgA (a marker of neuroendocrine cells) was also evaluated using Western blotting (Fig. 3A), and CgA expression was significantly higher in NPs in the CRSwNP group than in UTs from the control group, but there was no significant difference in CgA expression in NPs between the ECRSwNP and NECRSwNP groups (Fig. 3B).
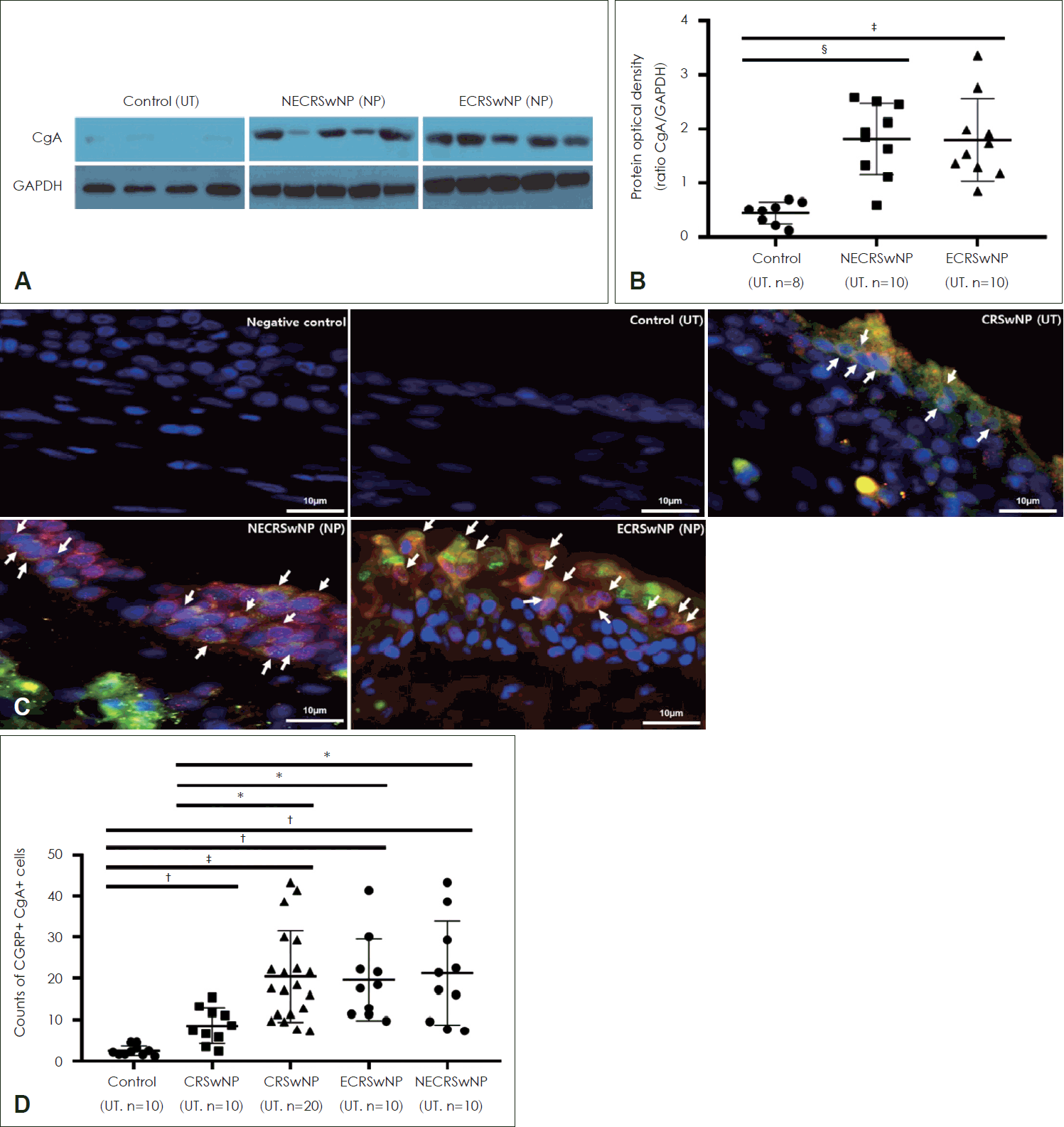
CGRP producing neuroendocrine cells were increased in nasal polyps of patients with CRSwNP. A and B: Expression of CgA protein levels in the tissues from UT of the control and NPs of the CRSwNP group. C: Representative image of immunofluorescence staining with CGRP and CgA (arrow, magnification, ×1000). Arrow indicates double positive cells of CGRP and CgA. D: The number of double positive cells (CGRP+ CgA+ cells) in the tissue among groups. Green: CgA, Red: CGRP. *p<0.05; †p<0.01; ‡p<0.001; §p<0.0001. UT, uncinated tissue; NECRSwNP, non-eosinophilic CRSwNP; NP, nasal polyp; ECRSwNP, eosinophilic CRSwNP; CgA, chromogranin A; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; CGRP, calcitonin gene-related peptide; CRSwNP, chronic rhinosinusitis with nasal polyp.
To identify CGRP-producing cells, double IF analysis targeting CgA and CGRP was conducted (Fig. 3C). The number of CgA+CGRP+ cells was significantly higher in tissues (UTs and NPs) from patients with CRSwNP than in UTs from controls (Fig. 3C and D). In the CRSwNP group, the number of CgA+CGRP+ cells was significantly higher in NPs than in UTs (Fig. 3D). There was no significant difference in cell counts between the ECRSwNP and NECRSwNP groups (Fig. 3D).
Exogenous CGRP on the expressions of various molecules in DNPCs
Cytokine arrays were conducted to elucidate the role of exogenous CGRP in the DNPCs of EPs (Fig. 4A). CGRP treatment reduced the expression of pro-inflammatory cytokines (e.g., IL-1β, IL-6, IL-17A) and angiogenesis-related molecules (e.g., angiogenin, angioprotein-1, endoglin). However, CGRP did not suppress the expression of tissue remodeling-related proteins (e.g., chitinase 3-like 1 [CHI3L1], macrophage migration inhibitory factor [MIF], trefoil factor 3 [TFF3]), and it increased the expression of anti-inflammatory cytokines (e.g., IL-1ra, IL-4, IL-10) (Fig. 4B).
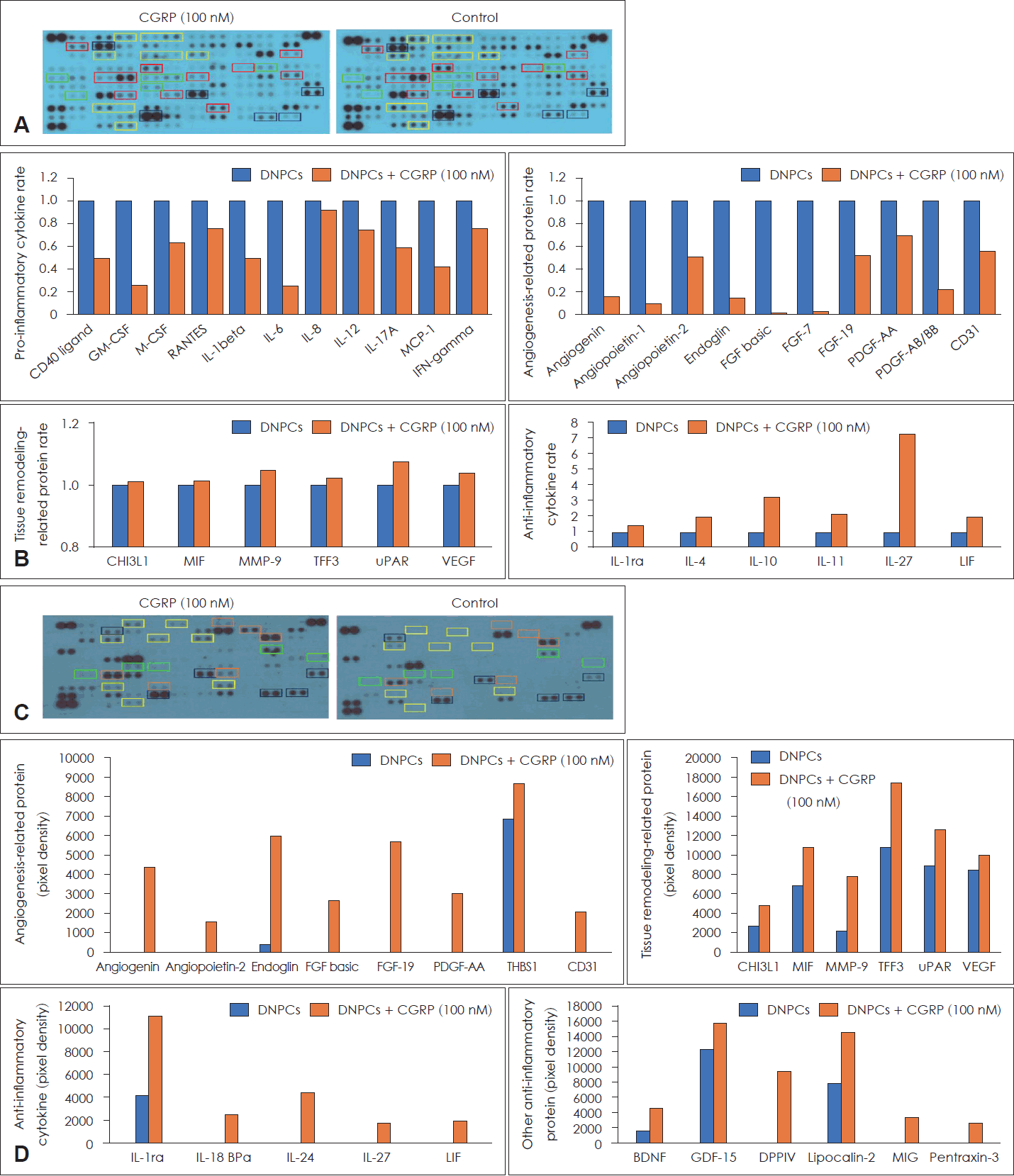
Expression of molecules by exogenous CGRP in DNPCs of eosinophilic- and non-eosinophilic NP. A and C: Expression of molecules by exogenous CGRP in DNPCs of eosinophilic NP (A) and non-eosinophilic NP (C). B and D: The orange bar indicates the ratio of the control group to the CGRP-treated group for each associated molecule in DNPCs of eosinophilic NP (B) and non-eosinophilic NP (D). Red boxes: pro-inflammatory cytokines, Green boxes: anti-inflammatory cytokines, Yellow boxes: angiogenesis-related proteins, Blue boxes: tissue remodeling-related proteins, Orange boxes: other anti-inflammatory proteins. CGRP, calcitonin gene-related peptide; DNPCs, dispersed nasal polyp cells; NP, nasal polyp; CD, cluster of differentiation; GM-CSF, granulocyte-macrophage colony-stimulating factor; M-CSF, macrophage colony-stimulating factor; RANTES, Regulated upon Activation, Normal T Cell Expressed and Presumably Secreted; IL, interleukin; MCP, monocyte chemoattractant protein; IFN, interferon; FGF, fibroblast growth factor; PDGF, platelet-derived growth factor; CHI3L1, chitinase-3-like protein 1; MIF, macrophage migration inhibitory factor; MMP3, matrix metalloproteinase-3; TFF3, trefoil factor 3; uPAR, urokinase-type plasminogen activator receptor; VEGF, vascular endothelial growth factor; LIF, leukemia inhibitory factor; THBS1, thrombospondin 1; BPa, binding proteins; BDNF, brain-derived neurotrophic factor; GDF, growth/differentiation factor; DPPIV, dipeptidyl peptidase-4; MIG, chemokine ligand 9.
The effects of exogenous CGRP were also evaluated in the DNPCs of N-EPs (Fig. 4C). The expression of molecules associated with angiogenesis (e.g., angiogenin, angioprotein-1, endoglin), tissue remodeling (e.g., CHI3L1, MIF, TFF3), and anti-inflammatory response (e.g., IL-1ra, IL-10, IL-27) was increased by treatment with exogenous CGRP in N-EPs (Fig. 4D).
CGRP suppressed the inflammatory response by inhibiting NF-κB signal pathways
The effects of CGRP on NF-κB expression were evaluated in THP-1 cells using Western blotting and ELISA. CGRP treatment inhibited IκB degradation, and thus, its expression was increased in the cytosol. CGRP also suppressed the translocation of NF-κB to the nucleus (Fig. 5A). LPS-induced IL-1β and IL-6 secretion was also significantly decreased by CGRP treatment in THP-1 cells (Fig. 5B and C).
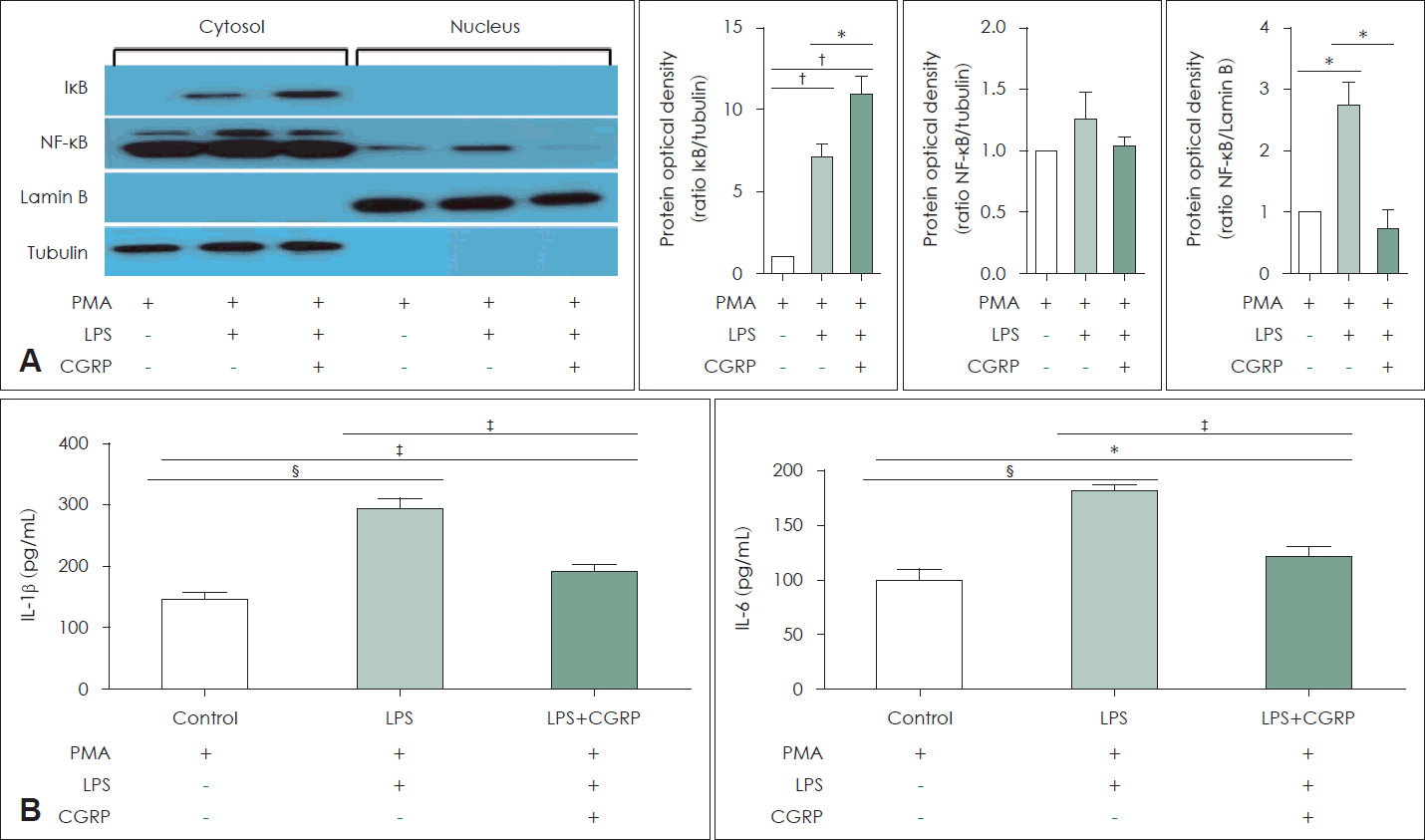
Exogenous CGRP was inhibited LPS-induced NF-κB activation in M1 macrophages. A: Expressions of NF-κB and I-κB protein levels were analyzed in the cytosol and nucleus using western blot assay in M1 macrophage. B: Productions of IL-1β and IL-6 levels were analyzed using ELISA. *p<0.05; †p<0.01; ‡p<0.001; §p<0.0001. IκB, nuclear factor of kappa light polypeptide gene enhancer in B cells inhibitor; NF-κB, nuclear factor-kappa light chain enhancer of activated B cells; PMA, phorbol 12-myristate 13-acetate; LPS, lipopolysaccharides; CGRP, calcitonin gene-related peptide; IL, interleukin.
Discussion
This study is the first in our knowledge to evaluate the CGRP expression in CRSwNP patients and see what kind of cytokines are increasingly expressed when stimulation with CGRP that results in anti-inflammatory effect. Neuropeptides have been found in nasal mucosa, and they are secreted by sensory nerves during inflammatory reactions [16]. In patients with rhinosinusitis, parasympathetic nerves are activated via sensory immunogenic activation in the V2 trigeminal nerve, that neuropeptides are secreted [17]. In the present study, CGRP expression was more strongly elevated in NPs from patients with CRS than in uncinated mucosa from the same patients. This shows the possibility that immunologic mechanisms regulating neural control maybe closely involved in CGRP production. It is known that CGRP expression in LPS-induced RAW 264.7 macrophages increases in a concentration- and time-dependent manner [18]. Tryptases and nerve growth factor (NGF) produced by mast cells are also known to stimulate sensory neurons or immune cells and induce the expression of CGRP under inflammatory conditions [19]. Few previous studies have shown that NGF strongly induces the expression of CGRP in activated B cells, and LPS stimulation increases the expression of NGF and CGRP in human monocytes [20,21]. This kind of immunologic mechanisms may be involved in the upregulation of CGRP in NPs, although experiments on CGRP production by these immune cells were not included in the current study.
Neuropeptides are also released from neuroendocrine cells. Neuroendocrine cells are specialized epithelial cells located in the upper and lower airway epithelia, intestines, and pancreas [22]. Mice with ovalbumin-induced asthma and patients with asthma exhibit a higher number of neuroendocrine cells than their control counterparts [9]. It has been reported that components such as neuroendocrine cells are present in the human nasal mucosa and distributed in submucosal glands and the lamina propria [23]. Neuropeptides released from these components are also involved in the regulation of immune responses, and dysfunction of neuropeptides acts as one of the pathogenic mechanisms of asthma, chronic lung disease, and allergies [24].
In this study, we aimed to evaluate the CGRP expression in CRSwNP patients and see what kind of cytokines are increasingly expressed when stimulation with CGRP. We attempted to elucidate the CGRP-secreting cells in NPs using CgA antibody. Most CGRP-secreting neuroendocrine cells were distributed along the mucosal surface of NPs, which is under more severe inflammation than UTs from patients with CRSwNP or control subjects. We conducted cytokine arrays to examine the effects of CGRP on the expression of various cytokines or molecules associated with various cellular responses, such as inflammatory and anti-inflammatory responses, angiogenesis, and tissue remodeling. Indeed, CGRP affected the expression of various molecules in DNPCs. The inhibitory effects of CGRP were more prominent in DNPCs from EPs than in those from N-EPs.
However, there are limitations to this study. One limitation of this study is the insufficient number of patient samples, especially for the cytokine arrays. Also, the main molecules expressed in EP and N-EP differed that we chose to use different parameters when comparing the cytokine array results. It is possible that EPs collected in the present study exhibited more severe inflammation than N-EPs. Last but not least, we could not clarify on the contradicting results of N-EP and EP on the angiotensin related proteins. The limitations above may possibly resolve by using a larger number of samples in the future study.
As CGRP expression was linked to anti-inflammatory activities in NPs, we believed that CGRP was upregulated to compensate for mucosal inflammation in NPs. Therefore, we hypothesized that CGRP, especially α-CGRP, could serve as a biomarker for disease severity. The reason why only α-CGRP expression was significantly correlated with the L-M CT score may be that β-CGRP was produced at lower levels than less than those of α-CGRP in NPs. It has been reported that both α- and β-CGRP have similar biological functions, and their expression is not correlated, although α-CGRP is known to be produced and released in the central and peripheral nervous systems whereas β-CGRP is released in the gastrointestinal tract and immune system [25].
It has been reported that CGRP inhibits the phosphorylation of IκB in RAW 264.7 cells, thereby inhibiting the activity of NF-κB and reducing the expression of cytokines [12]. The reduced activity of NF-κB following CGRP expression in vitro and in vivo has been examined using transgenic NF-κB luciferase reporter mice [26]. LPS-induced expression of CCL2, CXCL1, and CXCL8 was decreased in HMEC-1 and pHDMEc cells by inhibiting the activity of NF-κB using CGRP [27]. To verify the CGRP-mediated inhibition of NF-κB signaling, M1 macrophages, which were differentiated from the THP-1 human monocyte line by stimulation with PMA, were used. The phosphorylation of IκB induced by LPS was inhibited by CGRP, which reduced the translocation of NF-κB from the cytosol to the nucleus. This treatment also reduced the LPS-induced expression of pro-inflammatory cytokines such as IL-1β and IL-6, which are produced through the NF-κB pathway. We only investigated the NF-κB signaling pathway, but other signaling pathways associated with the anti-inflammatory effects of CGRP also may be needed to verify in the future.
In conclusion, the expression and anti-inflammatory effects of CGRP indicate that it may play a pivotal role in nasal polypogenesis and the modulation of mucosal inflammation. Future studies are needed to evaluate this possibility on the effect of CGRP in the pathogenesis of CRSwNP.
Acknowledgements
None
Notes
Author Contribution
Conceptualization: Yong Min Kim. Data curation: Seung Hyeon Choi. Formal analysis: Yong Min Kim, Soo Kyoung Park, Seulgi Lee, Jaein Chung. Investigation: Min Gyu Kim, Se Yeon Im. Methodology: Yong Min Kim, Soo Kyoung Park, Ki-Sang Rha. Project administration: Yong Min Kim. Supervision: Yong Min Kim, Ki-Sang Rha. Validation: Yong Min Kim, Seung Hyeon Choi. Visualization: Seulgi Lee, Jaein Chung. Writing—original draft: Jaein Chung, Seulgi Lee. Writing—review & editing: Yong Min Kim, Jaein Chung.