우연성 부비동염과 대사증후군 간의 상관 관계: 강북삼성병원 건강검진센터 자료를 바탕으로 한 단면 코호트 연구
Association Between Metabolic Syndrome and Incidental Paranasal Sinusitis: A Cross-Sectional Cohort Study of Kangbuk Samsung Health Care Center
Article information
Trans Abstract
Background and Objectives
Metabolic syndrome is a medical problem featured by the coexistence of several risk factors such as, heart disease, stroke, and diabetes. The association between metabolic syndrome and incidental paranasal sinusitis has not been definitely proved yet. This study was established to prove if metabolic syndrome might be related to incidental paranasal sinusitis.
Subjects and Method
Of 5682 patients who had undergone brain MRI for regular checkups between 2011 and 2018 at Kangbuk Samsung Health Care Center, we analyzed 2882 cases with normal sinus and incidental paranasal sinusitis that had been deciphered by specialized radiologists. Data of various components of metabolic syndrome were also used. Multivariable logistic regression analysis was performed to calculate the odds ratio (OR) of having incidental paranasal sinusitis between subjects with metabolic syndrome components and those without.
Results
Of 2882 patients aged over 18 years (mean age, 51.8±9.47 years; 55.4% male), 353 incidental paranasal sinusitis subjects (232 male with mean age of 51.39±9.27 years and 121 female with mean age of 52.43±9.69 years) were identified. Metabolic syndrome was identified in 554 patients. The adjusted OR of having incidental paranasal sinusitis was 3.03 (95% confidence interval [CI]: 1.58-5.83) in female with metabolic syndrome. In female, the adjusted OR of having incidental paranasal sinusitis was 2.10 (95% CI: 1.20-3.67) in those with low high density lipoprotein (HDL) cholesterol (<50 mg/dL) and 1.83 (95% CI: 1.06-3.16) in those with higher body mass index (BMI) (≥25 kg/m2).
Conclusion
Results of this study suggest that female with metabolic syndrome, low HDL cholesterol, and high BMI have higher risks for incidental paranasal sinusitis.
Introduction
Incidental paranasal sinusitis has been increasingly reported because of the diagnostic use of brain MRI for head and neck disease is becoming more frequent [1,2]. It might be expected that if incidental paranasal sinusitis detected earlier, the faster the management could be taken. However, clinical causes of incidental sinusitis remain unknown [3-7]. Asymptomatic incidental sinusitis patients have been mostly regarded as nonsignificant cases [3]. Several recent studies have revealed that diabetes and body mass index (BMI) are associated with incidental sinusitis [8,9]. It has also been shown that hypertension is associated with incidental sinusitis in female [10].
Metabolic syndrome is a group of components resulting from overnutrition, sedentary modern lifestyle, and excessive adiposity [11]. The prevalence rate of metabolic syndrome in Korea has increased continuously from 24.9% in 1998 to 31.3% in 2007 [12,13]. It has become to the fore in the modern society because metabolic syndrome is well known to increase the risk of cardiovascular diseases and type 2 diabetes [11,12]. Metabolic syndrome is diagnosed when there are three positive results for the following five criteria: high waist circumference, high blood pressure (BP), high triglyceride (TG), high fasting glucose, and low high density lipoprotein (HDL) cholesterol [12,14].
As described earlier, diabetes, BMI, and hypertension as components of metabolic syndrome criteria all have been reported to be associated with incidental paranasal sinusitis. However, the association between metabolic syndrome and incidental paranasal sinusitis has not been definitely proved yet. Thus, the purpose of this study was to clarify if metabolic syndrome might be related to incidental paranasal sinusitis.
Subjects and Methods
Study designation and study participants
This was a cross-sectional cohort study analyzing data obtained from regular health checkups performed at Kangbuk Samsung Medical Center, Seoul, Korea to determine the association between metabolic syndrome and incidental paranasal sinusitis. Of a total of 521848 patients with regular health checkups between 2011 and 2018 at Kangbuk Samsung Health Care Center, 5682 patients had undergone brain MRI. Of these 5682 patient, 2882 cases with normal sinus and paranasal sinusitis deciphered by specialized radiologists were analyzed. Data for components of metabolic syndrome were also used. All the patient’s data were obtained by internationally standardized questionnaire survey and body compositions such as height and weight were measured by trained nurses with the participants wearing a lightweight hospital gown and no shoes. Blood sampling was done by highly trained nurses and clinical laboratory scientists with over 8 hours fasting state. This study had some strengths, in terms of its large sample size, cohort design, detailed and standardized clinical and laboratory data. This study was reviewed and acknowledged by the Institutional Review Board (IRB) of Kangbuk Samsung Hospital (approval number: 2020-11-013). The requirement for informed consent was exempted by the IRB because only deidentified data collected during health-screening processes were used.
Metabolic syndrome as an independent variable
Metabolic syndrome was defined following the criteria of NCEP-ATP III, reflecting the waist measurement standard for Koreans. Detailed diagnostic criteria of metabolic syndrome are shown in Table 1 [12]. Following these criteria, subjects with three or more positive index above criteria were determined as metabolic syndrome group.
Paranasal sinusitis as a dependent variable
Brain MRI was performed using a 1.5-tesla scanner (SIGNA HDxt 1.5T; GE Healthcare, Tokyo, Japan). These images were deciphered by educated radiologists who did not know this research. Incidental paranasal sinusitis was defined as the existence of official readings by radiologists including sinusitis in at least one paranasal sinus.
Statistical analysis
Baseline characteristics of study participants are presented as mean±standard deviation for continuous variables and number (percentage) for categorical variables. Continuous variables were compared using student’s t-test. Categorical variables were compared using chi-square test or Fisher’s exact test. For continuous variables having abnormal distribution, they were presented as median (IQR) and compared using Mann-Whitney U test. Graphical method (using histogram) was used for determining whether data had a normal distribution.
The association between metabolic syndrome and incidental paranasal sinusitis was then analyzed. Odds ratios (ORs) with 95% confidence intervals (CIs) of having incidental paranasal sinusitis were calculated using a multivariable logistic regression analysis. ORs and 95% CIs were adjusted for several steps. Model 1 was adjusted for age and sex. Model 2 was adjusted for age, sex, regular exercise, smoking status, and alcohol consumption. In model 3, the final model, white blood cell (WBC), high sensitivity C-reactive peptide (hsCRP), and allergic history were further adjusted in addition to those adjusted in model 2. Individual components of metabolic syndrome diagnostic criteria were also analyzed using multivariable logistic regression analysis. ORs and the 95% CIs were calculated after adjustment. Adjusted variables were the same as above. All statistical analyses were performed using STATA version (16.1) (StataCorp LP, College Station, TX, USA). p-values less than 0.05 were considered statistically significant.
Results
Baseline characteristics of study population
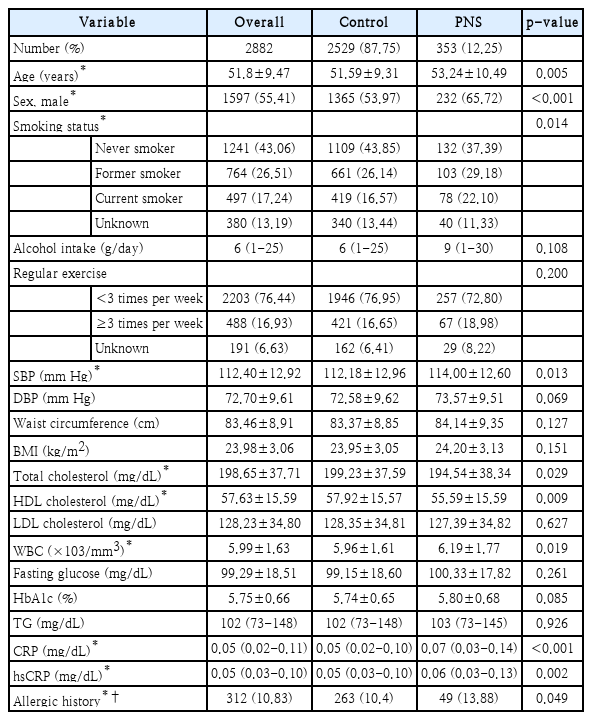
Of 2882 patients aged over 18 years (mean age, 51.8±9.47 years; 55.4% male), 353 subjects (232 male and 121 female; mean age, 53.24±10.49 years) were identified to have incidental paranasal sinusitis.
Table 2 shows baseline characteristic of participants, including age, sex, smoking status, alcohol intake, regular exercise, systolic and diastolic BP, waist circumference, BMI, total cholesterol, HDL cholesterol, low density lipoprotein (LDL) cholesterol, WBC count, fasting glucose, hemoglobin A1c, TG, C-reactive peptide (CRP), hsCRP, and allergic history. In the paranasal sinusitis group, age and proportion of male were higher compared to those in normal sinus group.
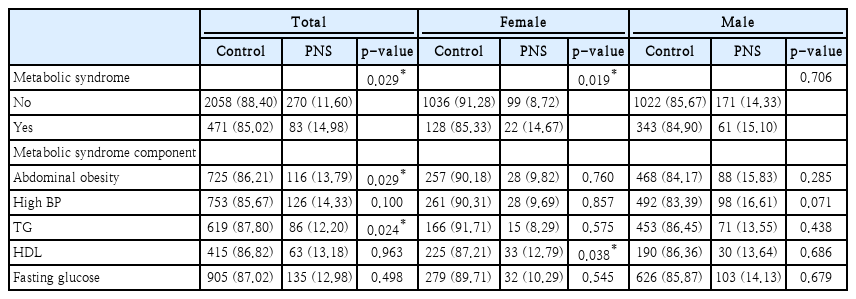
Prevalence comparison of metabolic syndrome and incidental paranasal sinusitis
Table 3 shows prevalence of metabolic syndrome and incidental paranasal sinusitis among total sex, female, and male group. Significant results were seen in total sex group (p=0.029) and female group (p=0.019). In male group, prevalence of metabolic syndrome was seemed high. However, no significant result was seen between metabolic syndrome and incidental paranasal sinusitis (p=0). Also, individual component of metabolic syndrome was analyzed in Table 3.
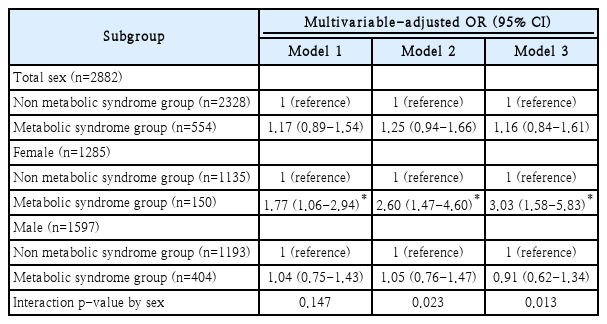
Association between metabolic syndrome and paranasal sinusitis according to sex
As shown in Table 2, age, sex, smoking status, systolic BP, total cholesterol, HDL cholesterol, WBC count, CRP, hsCRP, and allergic history are related to the prevalence of paranasal sinusitis. Supplementary Table 1 shows sex differences of baseline characteristics. Table 4 shows the association between metabolic syndrome and paranasal sinusitis. Three models were set according to the step-up adjustment. In multivariable logistic regression analysis, the adjusted OR was 1.77 (95% CI: 1.06-2.94) in model 1, 2.60 (95% CI: 1.47-4.60) in model 2, and 3.03 (95% CI: 1.58-5.83) in model 3 of the female group. These results indicated that higher OR was calculated if there were more adjustment variables in the female group. However, there was no significant tendency in the male group. There was no significant result in the total group either. Furthermore, the interaction p-value by sex were 0.023 and 0.013 in model 2 and model 3 respectively. It is assumed that because there were lots of sex differences of baseline characteristics in Supplementary Table 1, more significant results were elicited after step-up adjustment. In this model, relative excess risk due to interaction was -0.689 (95% CI; -1.747 to 0.370), attributable proportion due to interaction was -0.370 (95% CI; -0.975 to 0.235), and synergy index was 0.556 (95% CI; 0.247-1.249).
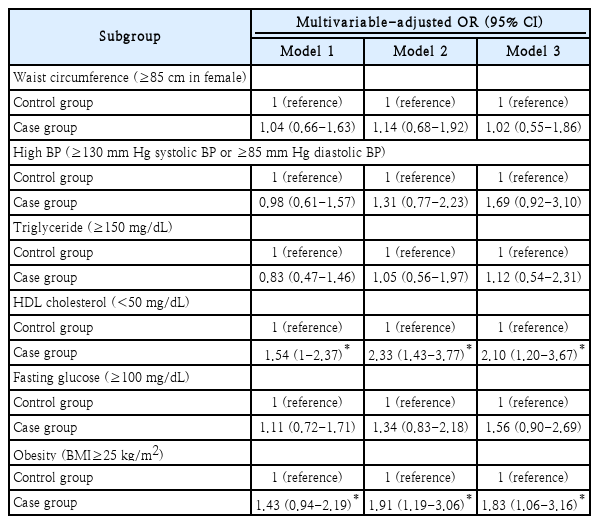
Association between individual components of metabolic syndrome diagnostic criteria and paranasal sinusitis in female
In succession, sub-analysis was performed to reveal if there were associations between individual components of metabolic syndrome diagnostic criteria and paranasal sinusitis. There were no significant results in the total sex group or the male group. However, in the female group, some components showed significant associations with paranasal sinusitis.
Table 5 shows associations between individual components of metabolic syndrome diagnostic criteria and incidental paranasal sinusitis in female. The association between BMI and incidental paranasal sinusitis in female was also found. The final adjusted OR was 2.10 (95% CI: 1.20-3.67) in those with low HDL cholesterol (<50 mg/dL) and 1.83 (95% CI: 1.06-3.16) in those with high BMI (≥25 kg/m2). However, waist circumference, high BP, TG, and fasting glucose did not show significant correlation with paranasal sinusitis.
Discussion
This large cross-sectional cohort study showed increases of incidental paranasal sinusitis in patients with metabolic syndrome. The association between incidental paranasal sinusitis and metabolic syndrome has been the subject of previous studies. One study showed that the prevalence of paranasal sinusitis was higher in patients with metabolic syndrome, especially in those with a high TG level, reduced high-density lipoprotein level, and elevated BP than in those without metabolic syndrome [15]. However, that study did not consider sex difference. Our study showed associations of paranasal sinusitis with low HDL (<50 mg/dL) and high BMI (≥25 kg/m2) in female. However, BP, TG level, and fasting glucose failed to show significant associations with paranasal sinusitis regardless of sex.
Another study has reported a significant positive association between dyslipidemia and paranasal sinusitis in patients with or without nasal polyp (adjusted OR: 1.36, 95% CI: 1.26-1.47, p<0.001) [16]. Both dyslipidemia and paranasal sinusitis are associated with chronic inflammation which has negative effects on the integrity of endothelial cells [16-18]. Dyslipidemia has been reported to be an important factor for oxidative stress causing endothelial dysfunction and atherosclerosis [16,19]. This oxidative stress has been found to be associated with the development of paranasal sinusitis [19].
Kabeya, et al. [8,9] have reported that patients with higher BMI are more likely to have paranasal sinus disease, consistent with our finding. They also reported a significant association of patients with diabetes and the prevalence of paranasal sinus disease (adjusted OR: 1.74, 95% CI: 1.12-2.71 in diabetic patients). Dales, et al. [10] have reported a correlation between hypertension and sinusitis in femlae (adjusted OR: 1.42, 95% CI: 1.04-1.95). However, fasting glucose and high BP showed no significant association with paranasal sinusitis in our study. The reason for such differences might be that numbers of patients analyzed in previous studies were relatively small. In addition, most studies had a cross-sectional design. To have more accurate results, a prospective study is needed.
Metabolic syndrome is associated with insulin resistance, adipose tissue dysfunction, dyslipidemia, especially high TG and low LDL levels [15]. This condition might be linked to abnormal levels of inflammatory markers such as cytokines and adipokines. Meanwhile, the correlation between essential hypertension and insulin resistance is already known [20]. Therefore, among diagnostic criteria of metabolic syndrome, dyslipidemia, elevated BP, and high fasting glucose level might cause chronic inflammatory conditions such as paranasal sinusitis.
Several hypotheses have been suggested for the development of paranasal sinusitis involving many environmental and host factors. ‘Immune barrier hypothesis’ is one of pathophysiologic explanations for paranasal sinusitis [21]. It assumes that chronic inflammation can cause defects in the mechanical barrier or innate immune response, making sinus mucosa become vulnerable to having paranasal sinusitis by microbes. As mentioned above, metabolically unhealthy patients, including those with dyslipidemia, elevated BP, high glucose level could show increased levels of inflammatory cytokines, inflammation-sensitive plasma proteins, and cellular adhesion molecules [16,22-24]. Consequently, these proinflammatory host factors could be linked to the development of paranasal sinusitis.
Strength of this study included its large sample size and randomized selection. As all data used in this study were from randomized regular health checkups, they were highly representative without a selection bias. Sex differences were seen in this study. This is not surprising as sex differences are well known for cardiovascular and immune diseases [10,25]. According to a previous study, female are more likely to have asthma [25] or smoking induced lung disease, suggesting that female show increased susceptibility to respiratory diseases [10,26-28]. Female might also have smaller sinus that could increase the risk of obstruction and paranasal sinus disease. Despite these explanations, further research is necessary to prove why metabolic syndrome and incidental paranasal sinusitis have association only in female.
This study has some limitations. First, paranasal sinusitis was determined according to incidental readings of brain image. Therefore, it might be unclear and controversial that there is the clinical relation of incidental findings detected in brain MRI scanning system. A previous study has shown that there is an association between paranasal sinus abnormality on MRI and symptoms of an upper respiratory infection [29]. However, some studies have suggested that there are differences between clinical symptoms and MRI findings [30,31]. Further research is needed to reveal the correlation between metabolic syndrome and clinically proved paranasal sinusitis. Secondly, brain MRI deciphering was done by several radiologists. Although they were highly trained experts, observer bias could not be completely excluded. Thirdly, the meaningful adjusted OR results were concluded only in female group.
In conclusion, this study suggests that female with metabolic syndrome, low HDL cholesterol, and high BMI are associated with incidental paranasal sinusitis. As for clinical appliance, otorhinolaryngologists and physicians should keep it in mind that patients with metabolic syndrome might have an increased risk of paranasal sinusitis. At the society level, metabolic syndrome is an increasing trend due to the modern lifestyle. It could lead to excessive healthcare expenditure. Therefore, it is necessary to make preventive effort for reducing the risk of paranasal sinusitis by taking regular sinus endoscopy examination or paranasal sinus X-ray check-up. More prospective studies are necessary to reveal the correlation between metabolic syndrome and paranasal sinusitis.
Supplementary Material
The Data Supplement is available with this article at https://doi.org/10.3342/kjorl-hns.2022.00500.
Acknowledgements
None
Notes
Author Contribution
Conceptualization: In Buhm Lee, Sang Hyuk Lee. Data curation: In Buhm Lee. Formal analysis: In Buhm Lee, Tae Hwan Kim, Jung Yup Lee. Investigation: In Buhm Lee, Kyung Chul Lee. Methodology: In Buhm Lee, Tae Hwan Kim. Project administration: Tae Hwan Kim, Sung Min Jin. Resources: In Buhm Lee, Tae Hwan Kim. Software: Mi Yeon Lee. Supervision: Sang Hyuk Lee. Validation: In Buhm Lee, Tae Hwan Kim, Sang Hyuk Lee. Visualization: In Buhm Lee. Writing—original drift: In Buhm Lee. Writing—review & editing: In Buhm Lee, Tae Hwan Kim, Sang Hyuk Lee.