국소 진행성 두경부암 환자에서 선행면역치료 후 수술적 치료: 수술 전후 합병증 및 치료 결과
Neoadjuvant Immunotherapy Following Definitive Surgical Treatment for Locoregionally Advanced Head and Neck Cancer: Perioperative Complication and Surgical Outcomes
Article information
Trans Abstract
Background and Objectives
We analyzed surgical outcomes, perioperative complications, and mortality in head and neck squamous cell carcinoma (HNSCC) in patients who underwent curative surgery following neoadjuvant immunotherapy.
Subjects and Method
The records of 36 HNSCC patients who underwent curative surgery with neoadjuvant immunotherapy and 69 HNSCC patients who received neoadjuvant chemotherapy were analyzed.
Results
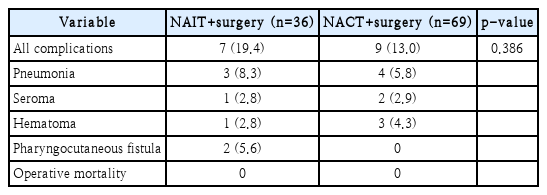
The average operation time was 315 minutes, and the average bleeding volume was 167 cc. The average length of hospital stay was 21 days. When evaluating surgical margin status, we found 24 patients (66.6%) who exhibited a negative margin. We found no case where surgery was impossible due to progression of the lesion during neoadjuvant immunotherapy. Compared to the neoadjuvant chemotherapy group, neoadjuvant immunotherapy group showed acceptable perioperative safety and complication profile. The postoperative complication rate was 19.4% in the neoadjuvant immunotherapy group and 13.0% in the neoadjuvant chemotherapy group (p=0.386). There were no serious complications during the recovery period after surgery or instances of death due to complications.
Conclusion
In HNSCC patients, there was no increase in the incidence of complications or mortality related to curative surgery after neoadjuvant immunotherapy.
Introduction
Head and neck squamous cell carcinoma (HNSCC) accounts for 90% of all head and neck cancers and is the sixth most common carcinoma worldwide [1]. Although smoking and drinking are the main causative factors, the incidence of HNSCC related to human papilloma virus (HPV) infection has been increasing. Despite advances in treatment modalities over the past few decades, the five-year survival rate of HNSCC patients persists at 40% to 60%, and many patients suffer from reduced swallowing and speech function after treatment [1,2]. Treatment methods for HNSCC are determined in accordance with the location and stage of the tumor, and surgery, radiotherapy, and chemotherapy are the mainstay treatment options. In the case of early HNSCC, surgery or radiation therapy can be performed alone. In patients with advanced HNSCC, combination therapy with surgery and chemoradiotherapy is essential; however, 40% of patients advanced eventually die due to disease relapse. Accordingly, it is necessary to develop innovative treatment strategies to overcome the dismal prognosis of HNSCC.
Recently, molecular targeting agents that focus on checkpoint proteins expressed in immune cells and tumor cells have been developed and are receiving attention [2]. Pembrolizumab and nivolumab are monoclonal antibodies targeting the programmed death protein 1 (PD-1) check protein, and have been approved by the Food and Drug Administration (FDA) for use in recurred or metastatic HNSCC [2]. Various clinical trials related to cancer immunotherapy are being conducted for carcinomas, including lung cancer, and studies on neoadjuvant immunotherapy for resectable solid tumors are underway [3,4]. However, there are not many studies on HNSCC, and very few have focused on treatment protocols that involve surgery after administration of neoadjuvant immunotherapy. Neoadjuvant immunotherapy is receiving attention as a new treatment to overcome the poor prognosis of HNSCC; however, research on how it affects morbidity or mortality in patients undergoing curative surgery after administration of neoadjuvant immunotherapy lacking. In this study, we aimed to analyze the treatment results of HNSCC patients who underwent curative surgery after neoadjuvant immunotherapy and to investigate the effects thereof on postoperative complications, mortality, and surgical outcomes.
Subjects and Methods
This retrospective study was approved by the Institutional Review Board (IRB), and, because it was conducted as a retrospective investigation, the IRB approved waived the need to obtain informed consent. This work was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans. From December 2018 to December 2020, a total of 36 HNSCC patients underwent curative surgery after neoadjuvant immunotherapy. The indications for neoadjuvant immunotherapy were as follows: 1) among malignant tumors in the oral cavity, hypopharynx, oropharynx, and larynx, only those histologically diagnosed as squamous cell carcinoma and only stage II-IVA HNSCCs were included in this study. The following were excluded: 1) patients with previous surgery or radiotherapy in the head and neck region, 2) those in whom surgery was not possible due to distant metastasis or invasion of vital organs at the time of diagnosis, 3) patients who were taking steroids, and 4) individuals with autoimmune disease or inflammatory disorders. To compare the safety and effectiveness of neoadjuvant immunotherapy and neoadjuvant chemotherapy, we performed the retrospective chart review of 69 patients with loco-regional advanced HNSCC who received neoadjuvant chemotherapy followed by surgery.
Our institute is conducting a phase II clinical trial to confirm the immunokinetics of durvalumab or durvalumab/tremelimumab as neoadjuvant therapy options for resectable HNSCC. The purpose of the present study was to investigate the safety of the treatment protocol by retrospectively analyzing the medical records of patients participating in the clinical trial and to assess the presence or absence of complications and surgical results in patients who underwent curative surgery following administration of neoadjuvant immunotherapy. The selection of patients receiving single (durvalumab-alone) or dual agents (durvalumab/tremelimumab-combination therapy) was randomized. Each patient received one cycle of the assigned drugs before surgery and underwent curative surgery within 2 to 8 weeks. Patients in the durvalumab-alone group received durvalumab (1500 mg) alone, and patients in the combined therapy group received durvalumab (1500 mg) and tremelimumab (75 mg) together; durvalumab is an anti–programmed death-ligand 1 monoclonal antibody, and tremelimumab is an anti-CTLA1 monoclonal antibody. After pathological examination of surgical specimens, the decision about whether to pursue adjuvant treatment or not was made with consideration of adverse pathologic factors, such as extranodal extension, surgical margin, lymphovascular invasion, and perineural invasion. All patients continued with drug use for up to 1 year after the end of treatment.
All patients underwent curative surgery. According to the preference of the operator or patient, a transoral robotic surgery or conventional surgical technique was performed. In all patients, selective or radical neck dissection was completed, depending on the presence or absence of cervical lymph node metastasis.
The patients’ personal information, surgical records, pathological results, and hospitalization records were collected and analyzed. During the perioperative period, complications related to surgery and mortality were investigated. Student’s ttest was used to compare continuous variables. To compare categorical variables, the chi-square or Fisher’s exact test was used. For statistical analysis, R version 4.04 (R Foundation for Statistical Computing, Vienna, Austria) was used. A p-value <0.05 was considered statistically significant.
Results
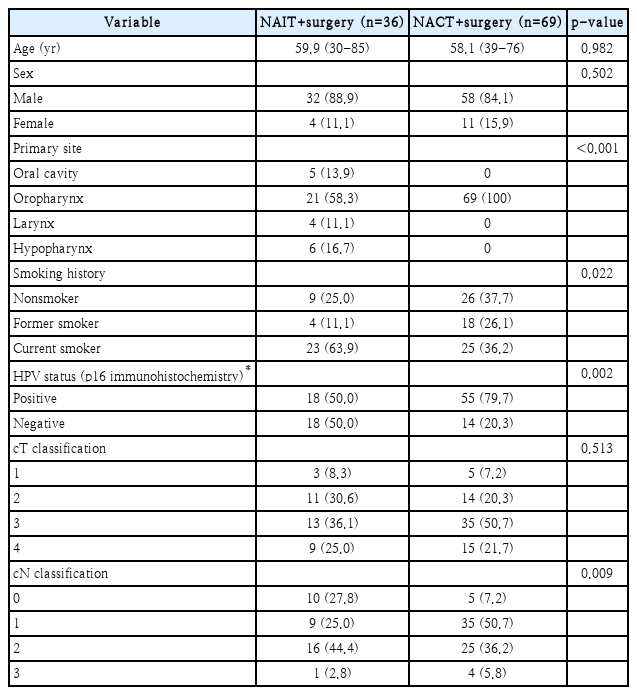
The mean age of the study participants was 59.9 years (range: 30-85 years), 32 were male, and 4 were female. The primary cancer site was the oral cavity in 5 cases, oropharynx in 21 cases, larynx in 4 cases, and hypopharynx in 6 cases. Twenty-five patients underwent transoral robotic surgery as a surgical method for primary lesions, and the remaining 11 patients underwent conventional surgery. Nine patients were nonsmokers, 4 were former smokers, and 23 were current smokers. HPV status was evaluated through p16 immunohistochemistry, and there were 18 positive and 18 negative cases, respectively. In regards to TNM stage, 3 patients were T1, 11 patients were T2, 13 patients were T3, and 9 patients were T4 cases. Regarding node classification, there were 10 patients with N0, 9 patients with N1, 16 patients with N2, and 1 patient with N3 status. Thirty patients underwent unilateral neck dissection and 6 patients underwent bilateral neck dissection. Additional clinicopathological information for our patients is presented in Table 1.
Of a total of 36 patients, 16 received durvalumab and the remaining 20 patients received a combination of durvalumab and tremelimumab. Clinico-pathologic factors, including age, sex, smoking, primary site, p16 status, TNM stage, lymphovascular invasion, perineural invasion, and extracapsular spread showed no significant differences between the groups. Also, no significant differences were observed in regards to neoadjuvant immunotherapy and immune-related adverse events between the groups.
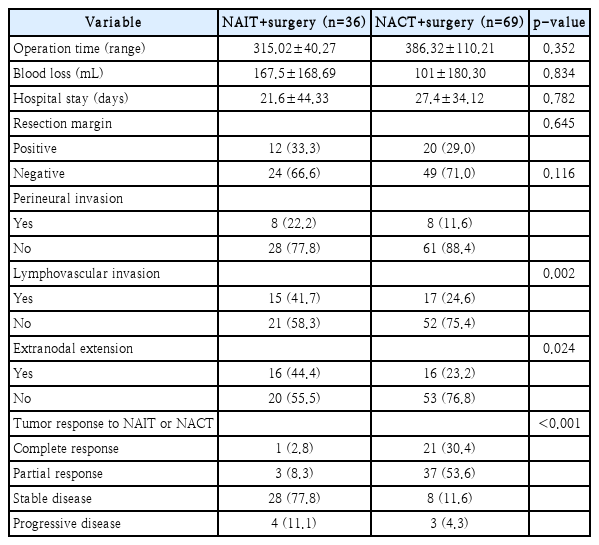
The average operation time was 315 minutes, and the average bleeding volume was 167 cc. The average length of hospital stay was 21 days. Twelve patients (33.3%) had a positive margin, and 24 patients (66.6%) had a negative margin. When cancer cells were observed in the surgical resection margin, we defined it as positive margin (n=9). Also, cases (n=3) in which the safety margin was less than 1 mm were defined as a close margin and classified as a positive margin. Perineural invasion was observed in 8 patients (22.2%), and lymphovascular invasion was observed in 15 patients (41.7%). Further, extranodal extension was observed in 16 patients (44.4%). Other operative outcomes and pathologic features were summarized in Table 2. One patient had a complete response after neoadjuvant immunotherapy, with cT3 tonsil cancer and negative p16 immunohistochemistry. The patient did not receive additional radiation therapy and was disease-free at the last follow-up. In the patient with a complete response after neoadjuvant immunotherapy, necrosis of 60% of the tumor mass and fibrosis of 40% of the tumor mass were observed, with no remaining viable tumor cells. Three patients had partial responses after neoadjuvant immunotherapy. In these 3 patients, the tumor mass was composed of 80%-95% viable tumor cells, 5%-10% necrosis, and 0%-10% fibrosis of the tumor mass. Among patients receiving neoadjuvant immunotherapy, 28 showed stable disease during neoadjuvant immunotherapy, and three experienced a partial response. Four patients showed progressive disease during the treatment period but maintained a resectable status and underwent curative surgery within 4 weeks. There was no case in which surgery was impossible due to hyperprogression of the lesion after neoadjuvant immunotherapy. The confirmation of pseudoprogression can be made by retrospective image analysis, resulting in premature cessation of immunotherapy. Biopsy can be used to deduce pseudoprogression before retrospective image analysis, but it is an invasive method that sometimes requires general anesthesia in head and neck cancer. Although some patients showed disease progression despite the administration of neoadjuvant immunotherapy in our study, all patients completed neoadjuvant immunotherapy without interruption of it and proceeded with the planned surgery. The postoperative complication rate was 19.4%, with three cases of pneumonia, one case of seroma, one case of hematoma, and two cases of pharyngeal fistula. There was no case of death due to complications.
During the study period, 69 patients with loco-regionally advanced HNSCC received neoadjuvant chemotherapy followed by surgery. We analyzed the difference between the neoadjuvant immunotherapy and neoadjuvant chemotherapy groups (Table 1). In the neoadjuvant chemotherapy group, most patients had oropharyngeal cancer and p16 positive status. However, the complication rate of the neoadjuvant chemotherapy group was 13.0%, and a positive resection margin reported in 25% of the neoadjuvant chemotherapy group. No significant differences were observed between the two groups in regards to perioperative morbidity and surgical outcomes (Table 3). Based on these results, we confirmed that neoadjuvant immunotherapy followed by surgery can be performed without adverse perioperative morbidity and surgical outcomes. However, due to the small numbers of patients enrolled in the study, it would be difficult to generalize these results, further study with large cohorts is warranted.
Among 36 patients, 24 were able to secure sufficient safety margin, and there was no case of any change in the original surgical plan after neoadjuvant immunotherapy. Thirteen patients were under follow-up without adjuvant treatment because no adverse pathologic factors were found after surgery. The remaining 23 patients underwent adjuvant radiation or chemotherapy. Among 23 patients who received adjuvant treatment, 22 received additional treatment within two months without delay after surgery, and one patient underwent adjuvant treatment five months later due to complications after surgery, with distant metastases ultimately identified during the follow-up period. There were four cases of recurrence during the study period, including one case of local recurrence, two cases of regional recurrence, and one case of distant metastasis. Two patients died, one due to disease progression and the other due to other concomitant medical disease.
Discussion
In a previous clinical trial, when nivolumab, an anti-PD-1 monoclonal antibody, was administered to HNSCC patients with recurrent or metastatic disease after platinum chemotherapy treatment, their overall survival was significantly increased relative to that of patients who received standard, single-agent treatment [4]. After this report, the FDA approved the use of anti-PD-1 antibody in patients with recurrent or metastatic HNSCC. With growing reports of the promising effects of immunological checkpoint inhibitors, interest in neoadjuvant treatment using the drug to overcome the dismal prognosis of HNSCC has grown. Clinical trial results for neoadjuvant immunotherapy in various carcinomas have already been reported, and clinical evidence supporting the use of neoadjuvant immunotherapy in resectable solid tumors has accumulated [5-8]. Neoadjuvant immunotherapy offers several advantages: it is possible to reduce the tumor volume and downstage before surgery, to evaluate pathologic response to the drug through examination of the surgical specimen, and to control micro-metastasis before curative surgery. However, there is also risk that the disease will progress during drug administration because definitive treatment must be postponed during this period. However, it has been reported in previous studies that a surgery delay of 3 to 4 weeks can be relatively safely in HNSCC patients [9-11]. Therefore, it is expected that neoadjuvant immunotherapy has the potential to bring about a breakthrough in the treatment of HNSCC.
Immune-checkpoint inhibitors including anti-PD1 or anti-CTLA4 monoclonal antibody promote the activation and expansion of T cells and elicit a wide range of immune-related adverse events in a variety of organs. The most common specific immune-related adverse events include diarrhea, colitis, pneumonitis, encephalitis, thyroiditis, interstitial nephritis, and skin rash. In this study, a total of eight treatment-related adverse events occurred during neoadjuvant immunotherapy, most of which were grade I or II, and there were no cases of treatment discontinuation or death due to serious complications. All patients underwent surgery within 2 to 8 weeks after administration of neoadjuvant immunotherapy, and there was no case in which surgery was impossible due to tumor progression. This result was consistent with previous reports of no cases of postponed surgery due to adverse events that occurred during the period of administration of neoadjuvant immunotherapy using anti-PD-(L)1 in HNSCC patients [12-14]. In the present study, during the perioperative period, the overall complication rate was 19.4%, and three cases of pneumonia, one case of seroma, one case of hematoma, and two cases of pharyngocutaneous fistula occurred. However, these complications were not correlated with the use of neoadjuvant immunotherapy. In all cases of complications, the patient recovered after medical or surgical treatment, and there were no deaths. The mean operation time was 315 minutes, the mean blood loss was 167 mL, and the mean hospital stay length was 21.6 days. No trends were observed in increased operating time, bleeding volume, or hospital stay due to neoadjuvant immunotherapy. After removal of the primary lesion through curative surgery, a sufficient safety margin was secured in 66.6% of cases, and four cases of disease recurrence and two cases of patient death occurred, one of which was due to other accompanying disease.
In treatment of HNSCC, curative surgery is performed after neoadjuvant therapy provides the following advantagesaIf tumor downstaging is achieved before curative surgery by reducing tumor volume, it is easier to secure sufficient safety margins during surgery. Based on the surgical specimen obtained after surgery, a biomarker study that can evaluate the response to the drug can be conducted, and it is possible to determine whether or not to administer adjuvant therapy after surgery by exploring important prognosis-related pathological factors. In addition, there is the potential to achieve control of micro-metastasis by administering systemic therapy before surgery. Recently, a treatment protocol using neoadjuvant chemotherapy and transoral robotic surgery has been reported, and satisfactory treatment results have been observed in patients with advanced head and neck cancer [15,16]. A new treatment protocol using neoadjuvant therapy and minimally invasive surgery has the potential to be revolutionary for HNSCC patients, and neoadjuvant therapy using immunotherapy may also induce sustained immunity to overcome the poor prognosis of HNSCC patients. However, there have been no studies on the impact of curative surgery after neoadjuvant immunotherapy on surgical outcomes or the occurrence of complications. According to a report by Bott, et al. [17], when immunotherapy is performed before surgery, fibrosis could be induced in the tumor and tumor microenvironment, and this can provoke technical challenges and perioperative issues during surgery. These findings can be problematic in head and neck cancer surgery since important cranial nerves are located in the neck, and preservation of these nerves is important to quality of life in HNSCC patients. In this study, neck dissection was performed in all patients, and there was no case of nerve sacrifice due to severe adhesions or fibrosis within the surgical field after neoadjuvant immunotherapy.
The limitations of our study are as follows: first, since this study was designed retrospectively, we should be careful in the interpretation of the results due to the potential for selection bias. Secondly, only small cohorts were enrolled, and it would be difficult to generalize our results to all head and cancer patients. Also, to investigate the effect of neoadjuvant immunotherapy on tumor microenvironment and clinical outcomes, we should analyze molecular profiles before and after neoadjuvant immunotherapy. Through future research, we expect to report immune pharmacodynamics using next generation sequencing technology to analyze specimens before and after treatment. Lastly, considering some complications such as fistula formation not found in neoadjuvant chemotherapy group occurred in neoadjuvant immunotherapy group, further research is needed on the effect of neoadjuvant immunotherapy on wound healing.
In conclusion, in patients with HNSCC after neoadjuvant immunotherapy, we observed no increase in the incidence of complications related to neoadjuvant immunotherapy before curative surgery or an increase in the number of deaths. It is believed that this treatment protocol can be applied relatively safely to manage HNSCC. Follow-up studies should be performed to identify molecular biomarkers for predicting patient response to immunotherapy as well as to verify the safety of the treatment protocol.
Acknowledgements
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (NRF-2021R1C1C1012004).
Notes
Author Contribution
Conceptualization: Young Min Park. Data curation: Da Hee Kim. Formal analysis: Se-Heon Kim. Funding acquisition: Hye Ryun Kim. Investigation: Yoon Woo Koh. Methodology: Young Min Park. Project administration: Da Hee Kim. Resources: Se-Heon Kim. Software: Hye Ryun Kim. Supervision: Yoon Woo Koh. Validation: Young Min Park. Visualization: Da Hee Kim. Writing—original draft: Ju Ha Park. Writing—review & editing: Yoon Woo Koh.