미세플라스틱과 두경부 건강: 이비인후과 질환 및 암 발생에 대한 새로운 시사점
Microplastics and Head and Neck Health: Emerging Implications for Otolaryngologic Disorders and Carcinogenesis
Article information
Trans Abstract
Microplastics (MPs), defined as plastic particles <5 mm in size, have become pervasive environmental contaminants with increasing relevance to human health. While originally investigated in marine environments, MPs are now routinely found in human tissues and fluids—including the blood, lungs, placenta, breast milk, and brain—demonstrating their widespread distribution and capacity for accumulation within the human body. The upper aerodigestive tract (UADT), comprising the nasal cavity, pharynx, larynx, and middle ear, serves as a key entry point for inhaled particles and is therefore particularly susceptible to MP deposition. Despite limited direct research on otolaryngologic tissues, mechanistic similarities with airborne pollutants such as particulate matter (PM2.5) suggest that MPs could elicit comparable biological effects, such as disruption of the epithelial barrier, induction of oxidative stress, mucosal inflammation, immune system disturbances, and fibrosis. These pathophysiological changes may play a role in the development of various ENT disorders, including chronic rhinosinusitis, allergic rhinitis, anosmia, and otitis media. Recently, there is increasing speculation that MPs could also contribute to head and neck (HN) carcinogenesis. MPs have been shown to transport adsorbed carcinogens and activate biological pathways including epithelial-mesenchymal transition (EMT), immune evasion, and remodeling of the tumor microenvironment, all of which are integral to tumor initiation, progression, and therapeutic resistance. Although causative associations have not yet been established, their detection in other organ systems susceptible to disease (e.g., lung, brain) further supports the possibility of oncologic implications. This review provides a comprehensive synthesis of existing data on MP exposure, accumulation in tissues, detection technologies, and the potential consequences for ENT health and malignancies of the HN. Drawing from toxicological, immunobiological, and oncological research, we highlight the critical need for systematic investigation of this underrecognized aspect of environmental risk in otolaryngology.
Introduction
The widespread presence of plastic pollution has introduced microplastic (MP) plastic particles less than 5 mm-as emerging environmental toxins of increasing concern for human health. Initially noted for their environmental effects, MPs are now being detected in a wide range of human biological matrices, including blood, placenta, breast milk, lung, liver, semen, and brain, which highlight their broad biodistribution and the potential for bioaccumulation [1-6]. These findings raise urgent concerns about the long-term consequences of chronic MP exposure, especially in vulnerable tissues, via mechanisms involving oxidative stress, chronic inflammation, immune dysregulation, and epithelial barrier compromise.
Among susceptible organ systems, the upper aerodigestive tract (UADT)-including the nasal cavity, pharynx, larynx, and middle ear-is particularly vulnerable to inhaled environmental particles. These anatomical regions function both as prime deposition sites for MPs and as essential centers for mucosal immunity and filtration. Alterations caused by particulate pollutants have been associated with otolaryngologic conditions such as chronic rhinosinusitis, allergic rhinitis, olfactory dysfunction, and Eustachian tube dysfunction. Given that MPs share similarities in size, chemical attributes, and immune-modulating capacity with particulate matter (PM2.5), it is conceivable that MPs could elicit similar-or potentially synergistic-effects in the pathogenesis of ENT diseases.
Moreover, recent mechanistic theories suggest that MPs may contribute to carcinogenesis in the head and neck (HN) region. Epidemiological studies have already demonstrated an association between PM2.5 exposure and an increased incidence of head and neck squamous cell carcinoma (HNSCC), especially with prolonged exposure periods pulmonology advisor [7]. MPs, which frequently transport adsorbed toxicants such as polycyclic aromatic hydrocarbons, heavy metals, and bisphenols, may facilitate mutagenesis, oxidative damage, immune dysregulation, and epithelial-mesenchymal transition (EMT)-all processes implicated in tumor development and resistance to therapy.
In this review, we consolidate existing evidence regarding systemic distribution, physicochemical profiles, and analytical methods for detecting MPs in human tissues, with particular attention to their implications for otolaryngologic disorders and HN malignancies. We examine proposed mechanistic pathways-including oxidative stress, persistent inflammation, barrier impairment, immune response alteration, fibrotic changes, and resistance to therapies-and detail comprehensive, multidisciplinary research approaches needed to substantiate these associations. Through the integration of perspectives from environmental toxicology, mucosal immunology, and oncologic research, this review seeks to highlight a vital, yet insufficiently explored, aspect of environmental health in otolaryngology and HN surgical practice.
Systemic Distribution and Bioaccumulation of MPs in Human Tissues
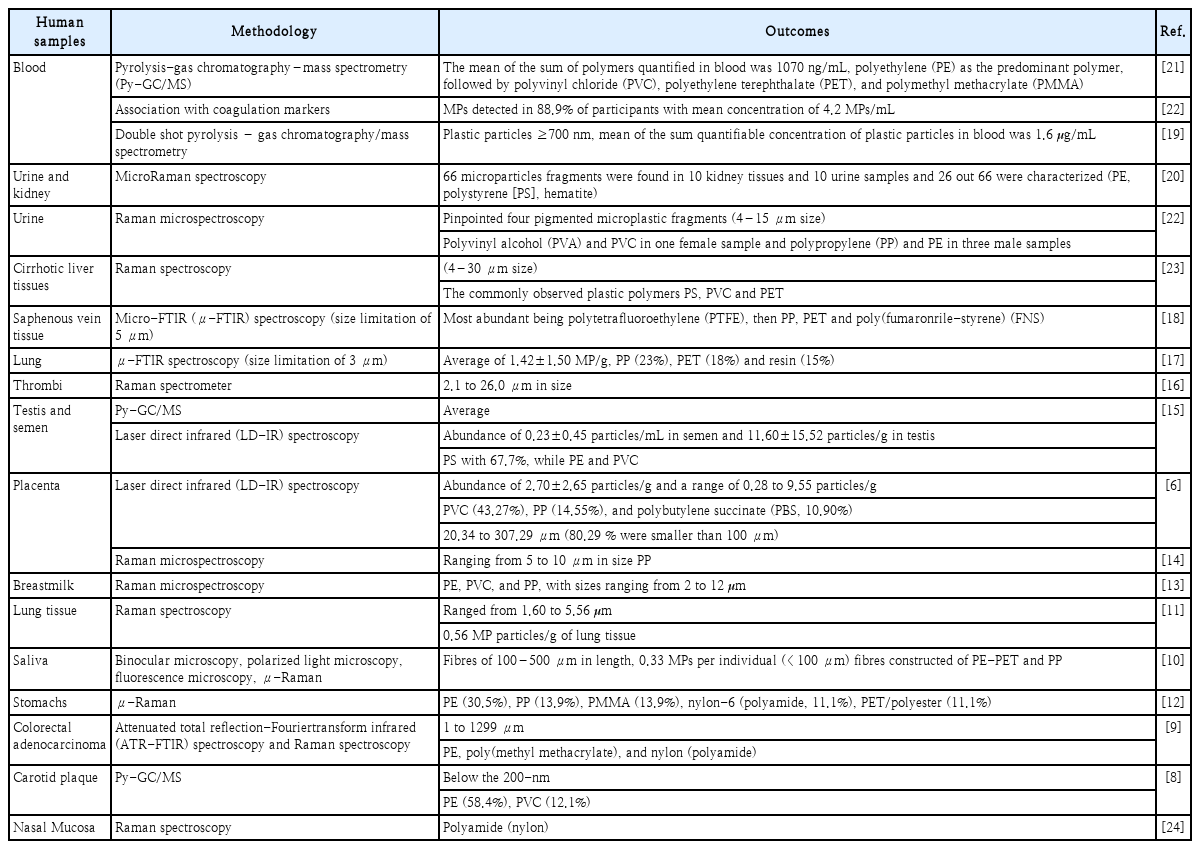
MP exposure has become an unavoidable aspect of the environment, with accumulating evidence confirming human exposure through multiple pathways and subsequent accumulation within tissues. Plastic particles in the micro-and nano-size ranges have now been identified in various human tissues and fluids-including blood, urine, feces, lung tissue, breast milk, semen, and placenta (Table 1) [6,8-24]-demonstrating systemic distribution and a significant potential for chronic bioaccumulation. The recurrent presence of MPs in placental tissue and meconium is of particular concern, as this suggests possible direct fetal exposure and raises significant concerns regarding developmental and transgenerational toxicity. While natural elimination pathways-such as fecal, urinary, and respiratory clearance-exist in humans, recent findings of MPs in postmortem tissues highlight the persistence of these particles and indicate that innate clearance mechanisms may be inadequate [25,26]. Studies of cadaver organs have demonstrated that MP concentrations in brain tissue may be up to 7-30 times higher compared to the liver or kidneys [1]. Additionally, levels of MPs identified in brain samples have risen by approximately 50% from 2016 to 2024, most likely reflecting the global escalation in plastic production and environmental dissemination [27]. Notably, individuals diagnosed with dementia have demonstrated up to tenfold higher concentrations of MPs in brain tissue than cognitively normal controls, suggesting a potential association between neuroplastic burden and neurodegenerative disease [28]. Moreover, patients with chronic diseases exhibit increased MP accumulation, particularly in lung, heart, and intestinal tissues [29,30]. These observations imply that disease-associated changes-such as persistent inflammation and tissue fibrosis-may enhance MP retention. Data from animal models further support the possibility of long-term harm; for instance, maternal exposure to polyethylene (PE) MPs during pregnancy has been shown to result in offspring exhibiting behaviors aligned with autism spectrum disorders [31,32]. Evidence for MPs in both the human placenta and neonatal meconium substantiates the likelihood of in utero exposure.
Collectively, the specific accumulation of MPs in the brain, increased tissue burdens in individuals with chronic disease, and the potential consequences for developmental and neurocognitive outcomes emphasize the urgent need to address MP exposure as a major public health issue that may have intergenerational repercussions.
Types and Characteristics of MPs Detected in the Human Body
A variety of MP polymers have been identified in human tissues, such as PE, polyethylene terephthalate (PET) [33], polypropylene (PP), polystyrene (PS) [34], polyvinyl chloride (PVC), and polycarbonate (PC) (Fig. 1) [35-41]. Within human populations, pregnant women and infants are recognized as groups with heightened susceptibility to MP exposure. Significantly, infant fecal samples reveal PET concentrations ranging from 5700 to 82000 ng/g (median: 36000 ng/g), about ten times higher than those measured in adult feces (2200-16000 ng/g; median: 2600 ng/g), a finding that suggests increased exposure during early developmental stages. In human breast milk, PVC and PP MPs in the 2-12 μm size range have been documented, while PE is most abundantly detected in brain tissue, indicating distinctive patterns of organ-specific accumulation [1,42]. Collectively, these observations highlight the necessity of analyzing both polymer composition and particle size for accurate assessment of MP toxicity across different tissues.
Systemic distribution of microplastics (MPs) in human organs and tissues. This schematic illustrates human tissues where MPs have been detected, including the brain, lungs, oral cavity (saliva), gastrointestinal tract (stomach, colon), reproductive organs (testis, placenta), and various biological fluids. Identified polymer types include polyethylene (PE), polypropylene (PP), polystyrene (PS), polyvinyl chloride (PVC), polyethylene terephthalate (PET), polymethyl methacrylate (PMMA), nylon, and polybutylene succinate (PBS).
Experimental studies select specific MP types to align with their research aims and to reflect typical environmental exposures. PS [34] is frequently used as a model MPs in laboratories because of its standardized physicochemical characteristics, broad commercial availability, and uniform size range. PS particles, with diameters from 0.1 μm to 100 μm, are commonly utilized in research on cellular uptake, cytotoxicity, and inflammatory mechanisms.
Environmental exposure models typically use PE MPs given their dominance in worldwide plastic production and waste streams. PE is especially suitable for studies focusing on gastrointestinal exposure. PP is primarily chosen for inhalation experiments, owing to the prevalence of PP fibers found in household air. Likewise, PET-MPs, often originating from plastic bottles, serve as common materials in oral exposure assessments, mirroring frequent dietary contact. To enhance detection in experimental systems, researchers employ fluorescently labeled MPs to visualize and quantify their biodistribution, cellular uptake, and elimination in real time. Use of fluorescent labels has further enabled detailed monitoring of MP clearance and accumulation in specific tissues, thereby aiding in the evaluation of biological impacts.
Detection Methods of MPs in Biological Samples
Detection of MPs in biological samples presents distinctive analytical challenges due to the complex nature of biological matrices and the typically low concentrations and small dimensions of target particles. Developing and refining detection methods for human tissues, fluids, and excreta requires careful attention to both sensitivity and rigorous contamination control. Stereomicroscopy-based visual inspection is commonly employed as an initial screening tool to assess larger MPs (>100 μm). Nonetheless, its use in biological matrices is constrained by limited specificity, susceptibility to operator bias, and the absence of polymer chemical identification capabilities. Fourier-transform infrared (FTIR) spectroscopy, especially when implemented as micro-FTIR (μ-FTIR) imaging, provides polymer-specific chemical analysis via vibrational fingerprinting and supports automated detection and localization of particles within tissue sections or digested samples [42]. This approach routinely identifies particles as small as ~20 μm, and certain instrumentation can achieve detection down to 10 μm. Raman microspectroscopy delivers higher spatial resolution than FTIR, with the capacity to identify particles as small as 1 μm. Its potential for analyzing biological samples is substantial, though interpretation is often complicated by marked tissue autofluorescence, necessitating optimized sample preparation and careful analysis. Scanning electron microscopy combined with energy-dispersive X-ray spectroscopy (SEM-EDS) enables detailed visualization and elemental analysis, facilitating the evaluation of particle morphology and inorganic components [43,44]. Despite this, SEM-EDS is unable to directly distinguish plastic polymers and is generally applied alongside complementary spectroscopic methods.
Robust contamination control is essential during the analysis of biological samples, as laboratory environments and containers are prone to plastic contamination. Maintaining data quality necessitates the exclusive use of glass or metal equipment, operation in clean air facilities, and the avoidance of plastic-based personal protective items. Routine use of blank controls helps monitor and quantify background contamination. Blood, tissue, and feces matrices are rich in lipids, proteins, and salts that can interfere with spectroscopic analyses through increased light scattering, protein fluorescence, or chemical background signals. Consequently, pretreatment processes-including enzymatic digestion, density-based separation, and filtration-must be carefully optimized to eliminate biological components while ensuring the structural integrity of MPs is preserved.
Recent advances in fluorescence-based detection have enhanced the visualization and quantification of MPs in biological samples. Nile red staining, which binds selectively to hydrophobic surfaces, facilitates fluorescence-based detection of MPs, especially when used alongside confocal microscopy [45-47]. Moreover, fluorescence lifetime imaging microscopy detects MPs by utilizing fluorescence lifetime signatures instead of relying solely on intensity, thereby increasing specificity in differentiating plastic types within complex tissue matrices. For detailed chemical identification and quantification, pyrolysis–gas chromatography–mass spectrometry (Py-GC/MS) remains widely acknowledged as the gold standard. This technique involves thermal decomposition of polymers, followed by analysis of their pyrolysis products to determine the type and concentration of plastics present. Time-of-flight secondary ion mass spectrometry and laser desorption/ionization mass spectrometry additionally provide surface-level chemical profiles, allowing detection of plastic additives and degradation byproducts [48-51]. While the majority of current methodologies identify MPs larger than 10-20 μm in biological samples, ongoing technological developments are focused on enhancing sensitivity to enable detection of submicron and nanoplastic particles.
Implications of MPs for Otolaryngologic Health and Disease
The UADT-including the nasal cavity, nasopharynx, oropharynx, larynx, middle ear, and Eustachian tube-is persistently exposed to inhaled airborne particulates, positioning it as a key region for MP accumulation. While direct research connecting MPs to otolaryngologic pathologies remains scarce, established insights into particle-driven inflammation and disruption of mucosal barriers suggest a plausible basis for investigating the effects of MPs on ENT tissues.
Inhaled MPs have been shown to trigger oxidative stress and the production of proinflammatory cytokines in respiratory epithelial cells, thereby compromising epithelial barrier integrity, promoting increased tissue permeability, and sustaining chronic inflammation [34,52-56]. Like particulate matter (e.g., PM2.5), MPs can adversely affect mucociliary clearance by interfering with ciliary architecture and function, reducing a principal defense mechanism of the upper airway [57-61]. Both in vitro and in vivo research has documented fibrotic alterations and changes in tissue remodeling attributed to recurrent MP exposure [32,62-66]. These findings underscore concerns regarding how MP exposure may contribute to the development of various ENT disorders. Notably, persistent mucosal inflammation and compromised clearance mechanisms may heighten susceptibility to allergic rhinitis, chronic rhinosinusitis, and persistent laryngitis. Prolonged exposure to nanosized plastics could disrupt the olfactory epithelium, leading to impaired olfactory function and potentially resulting in hyposmia or anosmia [67-69]. Additionally, disturbance of Eustachian tube function from mucosal irritation and modified mucus viscosity could make individuals, especially children, more prone to otitis media.
Exposure to MPs may also influence innate and adaptive immune responses within ENT tissues. Evidence indicates that macrophages ingesting MPs display diminished phagocytosis and increased release of proinflammatory factors, possibly compromising mucosal immune defense [70-77]. Moreover, contact of airway epithelial cells with synthetic polymers has been found to alter cytokine secretion profiles, thereby affecting immune cell recruitment and activity in the airway mucosa. Certain groups could be disproportionately affected by MP exposure. Children, who are undergoing maturation of mucosal and immune barriers, as well as older adults, who tend to have diminished mucosal defensive capacity and ciliary motion, may be particularly at risk. Furthermore, those with underlying respiratory or allergic diseases might experience worsened symptoms due to MP-driven inflammation or disruption of barrier integrity [33,78-82].
Although direct investigations assessing MP accumulation in otolaryngologic tissues are presently unavailable, the combination of supporting biological rationale and indirect data from respiratory and neurological studies emphasizes a critical need for dedicated research in this area. Future research should focus on elucidating the deposition characteristics, cellular reactions, and possible clinical consequences of MPs in nasal, pharyngeal, laryngeal, and middle ear tissues, especially among individuals at greatest risk.
The development of minimally invasive detection methods for MPs in the HN region represents a critical frontier in environmental health monitoring. Recent advances in surface-enhanced Raman scattering (SERS) technology offer unprecedented sensitivity for molecular detection, with enhancement factors reaching 106 to 1014, enabling single-molecule identification capabilities [83-85]. Future research strategies should focus on developing SERS-functionalized swab-based systems for non-invasive sampling from oral cavities, nasal passages, and throat regions, where cotton or synthetic swabs coated with plasmonic nanoparticles could provide direct, real-time MPs identification without complex sample preparation [86,87]. Additionally, needle-based SERS probes present opportunities for minimally invasive tissue sampling in suspected contamination sites, utilizing fiber-optic delivery systems integrated with SERS-active substrates for in-situ detection [88-90]. The molecular fingerprinting capability of Raman spectroscopy enables unambiguous polymer identification based on unique vibrational signatures, overcoming limitations of conventional microscopic methods in complex biological matrices [91,92]. These approaches could revolutionize clinical screening protocols by providing point-of-care diagnostics for MPs exposure assessment, particularly valuable given the HN region’s role as primary entry points for airborne and ingested contamination.
MPs and Head and Neck Cancer: Emerging Mechanistic Hypotheses and a Framework for Future Research
Although definitive causal evidence linking MPs exposure to head and neck cancer (HNC) remains unavailable, accumulating mechanistic and environmental toxicology data support a conceptual model in which MPs could facilitate both carcinogenesis and the progression of established cancers. First, mechanistic similarities to established carcinogens, particularly ambient fine PM2.5, provide a strong rationale for hypotheses regarding carcinogenic potential. Similar to PM2.5, MPs are implicated in the induction of oxidative stress, DNA damage, and persistent inflammation, as well as the disruption of epithelial barriers and modulation of the immune response. Epidemiological studies have demonstrated associations between chronic PM2.5 exposure and an elevated risk of HNSCC, especially in areas characterized by high pollutant levels [93-95], and MPs frequently bearing adsorbed carcinogens such as polycyclic aromatic hydrocarbons, heavy metals, and bisphenols-may have analogous pro-oncogenic effects. Second, sustained inflammation and the establishment of a tumor-promoting microenvironment represent critical mechanisms of concern. MPs have been documented to increase the production of proinflammatory cytokines, including interleukin (IL)-6 and tumor necrosis factor-alpha, as well as elevate reactive oxygen species (ROS), leading to persistent mucosal inflammation and conditions favorable to malignant transformation [73,96]. This is particularly significant in the UADT-including the oropharynx, larynx, and nasal cavity-where mucosal tissues are consistently exposed to inhaled particles and microbial agents. Third, the potential for selective accumulation of MPs in tissues susceptible to malignant transformation is biologically plausible. While MPs have not yet been detected in HNC tissue specimens, their presence has been confirmed in human lungs, placenta, and even brain tissue [14,97], supporting the possibility of similar deposition in HN mucosa, especially among individuals with compromised mucociliary clearance or elevated environmental exposure. Fourth, experimental studies have demonstrated that MPs exposure can promote fibrotic tissue remodeling, drive EMT, and induce immune suppression, all of which may facilitate tumor cell invasion, metastasis, and resistance to therapy [98]. MPs have been shown to stimulate the expression of EMT-associated genes and modify the composition of the extracellular matrix, while fostering macrophage polarization toward M2-like immunosuppressive states. Finally, MPs may impair anti-tumor immune responses and contribute to therapeutic resistance via mechanisms that include decreased antigen presentation by dendritic cells, increased secretion of immunosuppressive cytokines (such as IL-10 and transforming growth factor-beta), and enhanced activation of antioxidant pathways like NRF2, which can diminish the cytotoxic efficacy of chemotherapy and radiotherapy. Collectively, these findings indicate that MPs may influence not only the onset of HNC, but also its progression, invasiveness, and resistance to conventional treatments. Future research directions should incorporate histopathological detection of MPs in HNC tissues through advanced imaging and chemical analyses (such as μ-FTIR and Py-GC/MS), as well as inhalation-based preclinical exposure models and in vitro assays evaluating proliferation, EMT, and immune modulation in HNSCC cell lines. Epidemiological studies combining environmental exposure data with cancer incidence and clinical outcomes will be crucial to rigorously assess this emerging hypothesis. Although a direct association between MPs and HNC is currently hypothetical, the intersection of biological plausibility, mechanistic similarities to established carcinogens, and the anatomical vulnerability of ENT tissues highlight the urgent need for comprehensive, multidisciplinary research.
Conclusion
MPs have become a widespread environmental contaminant, with increasing evidence indicating human exposure and systemic accumulation in various tissues, including the respiratory and gastrointestinal tracts, placenta, brain, and potentially the UADT. Although definitive causal relationships between MPs and specific otolaryngologic diseases or HNC have not yet been empirically established, expanding mechanistic data from in vitro, in vivo, and environmental health research suggests several biologically plausible pathways of harm. These mechanisms comprise epithelial barrier disruption, oxidative stress, chronic inflammation, immune modulation, and fibrotic tissue remodeling, all of which are recognized contributors to carcinogenesis and disease progression in HNC. The particular anatomical and functional susceptibility of ENT structures, including the nasal, pharyngeal, laryngeal, and middle ear mucosa, to inhaled MPs further highlights the necessity of evaluating MPs as a potential risk factor in both inflammatory and neoplastic disorders of the HN (Fig. 2). Theoretical models and analogies to PM2.5 emphasize the importance of closing this knowledge gap through systematic research. Future studies that combine environmental exposure measurements, histopathological assessments, and molecular profiling in clinical samples will be essential for clarifying the potential role of MPs in HN pathophysiology. Understanding the health effects associated with MPs could offer new insight into the environmental determinants of ENT diseases and facilitate advancements in prevention, risk assessment, and targeted therapy in HNC.
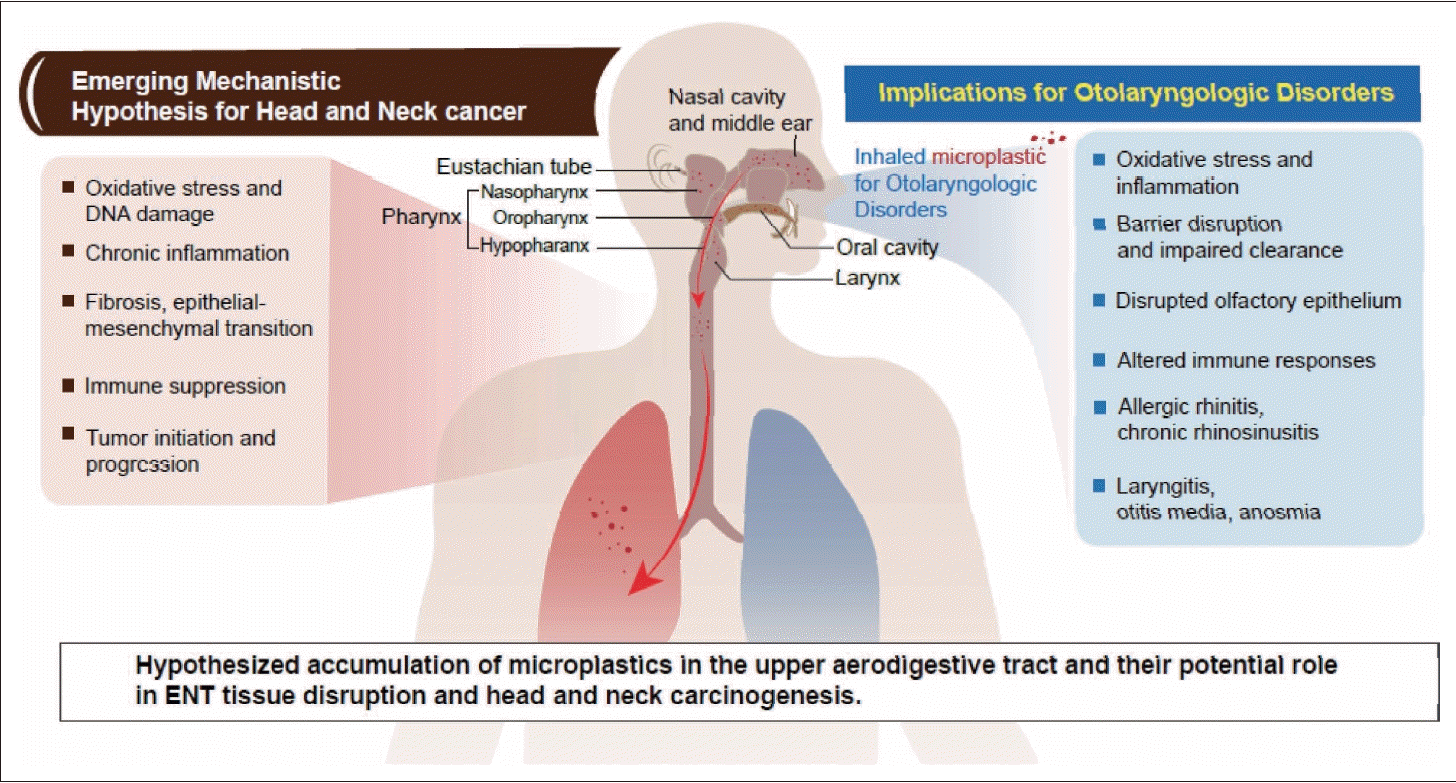
Hypothesized mechanistic impact of microplastics (MPs) on otolaryngologic disorders and head and neck cancer. MPs, through various exposure routes including inhalation and ingestion, are hypothesized to accumulate in the upper aerodigestive tract-encompassing the nasal cavity, middle ear, pharynx, and larynx-where they may provoke a range of pathological responses. The left panel outlines proposed mechanistic pathways by which MPs may contribute to head and neck carcinogenesis, including oxidative stress, chronic inflammation, epithelial-mesenchymal transition, immune suppression, and tumor initiation and progression. The right panel highlights potential implications for otolaryngologic health, such as epithelial barrier disruption, impaired mucociliary clearance, altered immune function, and heightened risk for diseases like allergic rhinitis, chronic rhinosinusitis, laryngitis, otitis media, and anosmia.
Notes
Acknowledgments
These works were supported by a National Research Foundation of Korea (NRF) grant (RS-2022-NR070495) and Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea.
Author Contribution
Conceptualization: Jun-Ook Park, Seung-Ah Yoo. Investigation: Jiyoung Yeo, Min-Kyung Nam. Writing—original draft: Min- Kyung Nam, Seung-Ah Yoo. Writing—review & editing: Jun-Ook Park, Seung-Ah Yoo.