박동성 이명을 주소로 내원한 경동맥-와우 피열: 증례 보고 및 문헌 고찰
Carotid Artery-Cochlear Dehiscence Presenting With Pulsatile Tinnitus: A Case Report and Review of the Literature
Article information
Trans Abstract
Carotid artery-cochlear dehiscence (CCD) is an extremely rare clinical entity defined as the dehiscence of the bony partition between the cochlea and the internal carotid artery (ICA). This condition may present with pulsatile tinnitus, hearing loss, or vertigo, mimicking other otologic and skull base pathologies. Moreover, an unrecognized CCD may lead to ICA injury during otologic procedures, particularly cochlear implantation. Here, we report the case of a 20-year-old male who presented with a 3-year history of right-sided exertion-induced pulsatile tinnitus without other otologic symptoms. He was diagnosed with CCD on non-enhanced temporal bone CT. As the first CCD to be reported in Korea, this case highlights the importance of recognizing this rare condition and underscores the diagnostic value of temporal bone CT.
Introduction
Pulsatile tinnitus is an auditory symptom that occurs in synchrony with the subject’s heartbeat without an external stimulus [1]. Although pulsatile tinnitus is less common than non-pulsatile tinnitus, accurate diagnosis is clinically important, as it affects up to 4% of the general population, and potentially treatable vascular etiologies have been identified in 44%-91% of cases [1].
Carotid artery-cochlear dehiscence (CCD) has rarely been reported through case reports and is described as a dehiscence of the bony partition between the petrous part of the internal carotid artery (ICA) and the cochlea [2,3].
This condition has two important clinical significances that neurotologic surgeons should be aware of [3]. First, this condition presents with symptoms such as pulsatile tinnitus, hearing loss (HL), and vertigo, which can mimic other otologic and skull base disorders, including otosclerosis, superior canal dehiscence syndrome (SCDS), glomus tumors, intracranial hypertension (HTN), aberrant ICA, sigmoid sinus dehiscence, and dehiscent jugular bulb [2,3]. Second, due to the potential risk of ICA injury during the procedure, recognition of the presence of CCD before otologic surgeries, especially cochlear implantation, is critical [3].
To the best of our knowledge, 12 cases have been reported worldwide, and none have been documented in Korea [2-12]. Here, we present the first case of CCD in Korea with an updated review of this condition.
Case
A 20-year-old nonobese male soldier presented to an outpatient clinic with a 3-year history of right-sided pulsatile tinnitus. The tinnitus was episodic, synchronous with his heartbeat, and typically triggered by physical exertion, especially aerobic exercises such as running. No additional otologic symptoms were observed. The patient’s medical and family histories were unremarkable.
Otoscopic examination of the right ear revealed normal findings. Thorough auscultation of the external auditory canal, mastoid, and neck using a stethoscope revealed no bruits or abnormal vascular sounds. Physical examinations, such as neck compression, neck rotation, and the Valsalva maneuver, were not performed as the patient did not complain of any symptoms during the outpatient visit. Pure-tone audiometry (PTA) demonstrated normal hearing bilaterally. Bilateral impedance audiometry revealed type A tympanograms. Pulsatile tinnitus of a vascular origin was suspected based on the characteristics of the symptoms. Non-enhanced temporal bone CT was performed to evaluate the bony structures of the temporal bone and their anatomical relationship with the surrounding vascular structures. Temporal bone CT revealed focal dehiscence of the bony partition between the basal turn of the right cochlea and the vertical segment of the right petrous ICA (Fig. 1). Additional imaging was not performed because the patient refused further evaluation.
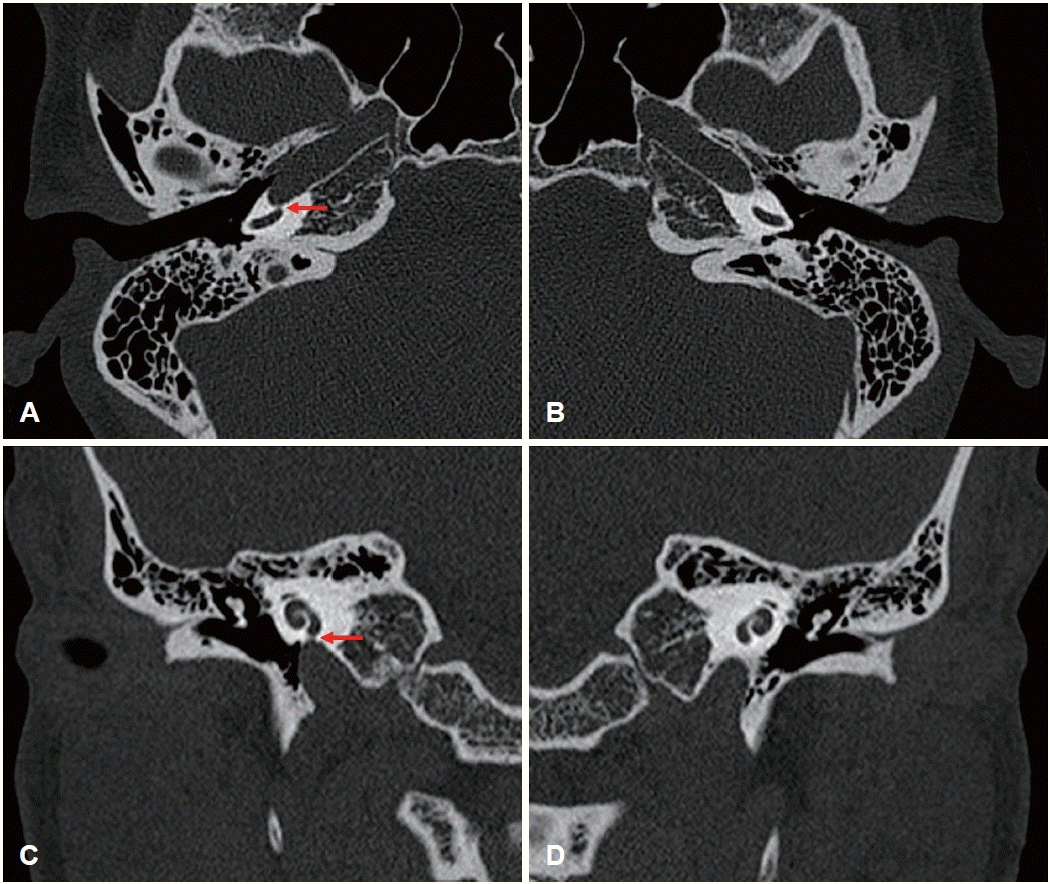
Non-enhanced temporal bone CT. Axial (A and B) and coronal (C and D) sections. Right side (A and C) and left side (B and D). Red arrows indicate focal bony dehiscence between the right cochlea and the right internal carotid artery.
Given the patient’s tolerable symptoms and the high surgical risks associated with the location of dehiscence, conservative management with close observation was recommended rather than surgical intervention.
Discussion
CCD is an extremely rare clinical entity, with only 12 cases reported worldwide since the first report by Modugno, et al. in 2004 (Table 1) [2-12]. Most previously reported cases have occurred in elderly patients and were associated with persistent symptoms, including HL and vertigo. In contrast, this case describes a young male with normal hearing and exertion-induced pulsatile tinnitus without vestibular symptoms. This distinct clinical presentation broadens the clinical spectrum of CCD.
In CCD, various types of HL have been observed on PTA. The most common type of HL was sensorineural HL (SNHL), which seems to be a result of chronic pressure from the ICA, causing damage to the intracochlear hair cells adjacent to the dehiscent region [9]. Indeed, bony defect at the basal turn of the cochlea tend to present with mid- to high-frequency SNHL, while bony defect at the apical turn present with low-frequency SNHL [9]. A larger dehiscence size in bilateral CCD case was also associated with higher hearing threshold levels [9]. These findings support this proposed mechanism. However, some cases show no clear correlation between defect location and audiometric pattern [3,10]. Some patients, including our patient, showed normal hearing thresholds despite the presence of CCD [9,11,12]. These observations suggest that a certain threshold size of dehiscence may be required for arterial pressure from the ICA to be transmitted significantly to the cochlea to produce HL [9].
Conductive HL (CHL) shown in the CCD can be explained by the third mobile window effect [6,7,13]. The third window is thought to increase the air conduction threshold by dissipating the sound energy from the cochlea primarily at low frequencies [13]. Otherwise, third window also increases the difference of the impedance between scala vestibuli and scala tympani by lowering the impedance of the scala vestibuli, consequently decreasing bone conduction threshold at the low frequencies [13]. The presence of CHL with intact stapedial reflexes in CCD supports that the underlying mechanism is consistent with third window effect [4,5,8].
The differential diagnosis of CCD includes conditions associated with pulsatile tinnitus, such as a dehiscent jugular bulb (Fig. 2A), sigmoid sinus dehiscence (Fig. 2B), and SCDS (Fig. 2C) [1,14,15]. To date, three hypotheses have been proposed to explain the vascular mechanisms of pulsatile tinnitus [1]. First, hemodynamic changes can induce turbulent flow of the bloodstream, which can transmit vascular sounds to the auditory system [1]. Second, dehiscence of the bony wall surrounding vascular structures may allow vascular sounds to be transmitted to the auditory system [1]. Third, the presence of a third mobile window in the inner ear can abnormally transmit vascular sounds directly into the perilymph [1].
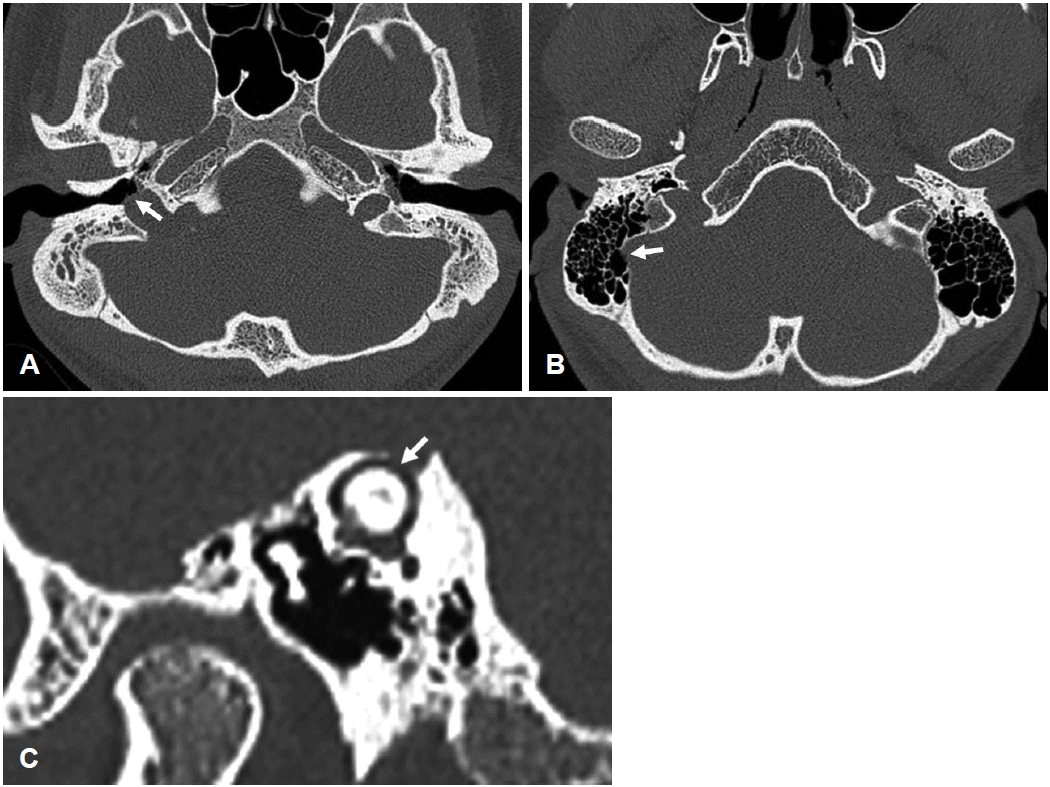
Non-enhanced temporal bone CT findings of diseases with bony dehiscence that can cause pulsatile tinnitus. A and B: Axial section. C: Reformatted image in the plane of superior semicircular canal. White arrows indicate sites of bony dehiscence. Dehiscent jugular bulb (A), sigmoid sinus dehiscence (B), and superior canal dehiscence syndrome (C). Reproduced with permission from Yang, et al. Korean J Otorhinolaryngol-Head Neck Surg 2016;59(1):68-72 [15].
In contrast to the first two mechanisms, SCDS and CCD are driven by a structural dehiscence that acts as a functional mobile window. As pressure fluctuations in the scala vestibuli can modify basilar membrane motion, cochlear microphonics can be altered in CCD, similar to SCDS [16]. Whereas CCD causes direct cochlear-vascular contact, vascular pulsations in SCDS are transmitted indirectly through cerebrospinal fluid on the vestibular side [14].
Clinically, CCD also shares several features with other third window lesions located in the vestibule, such as SCDS [13]. Similar to these conditions, CCD often presents with low-frequency CHL due to the third mobile window effect [13]. However, CCD is distinguished by the prominence of pulsatile tinnitus and by the occurrence of SNHL at frequencies adjacent to the site of dehiscence. Although relatively rare in CCD, vertigo induced by sound (Tullio phenomenon) or pressure changes (Hennebert’s sign) may also occur [11,14]. However, compared with other third window lesions, CCD often lacks vestibular symptoms, possibly because the third window is located relatively far from the vestibule [4]. In our patient, symptoms such as HL or vertigo were not observed. This may be explained by the patient’s relatively young age and the relatively small size of the dehiscence compared with previously reported cases.
Unlike CCD, dehiscence involving the jugular bulb or sigmoid sinus is located relatively far from the cochlea. Accordingly, vascular sounds of venous origin must pass through the surrounding media to reach the inner ear. Clinically, this often manifests as a relatively low-pitched sound, and its intensity may change with physical examinations such as neck compression, neck rotation, and Valsalva maneuver, providing useful points for differential diagnosis [1].
Although some differentiating points exist, these symptoms and audiological findings vary among cases and often overlap with those of other otologic and skull base pathologies. Consequently, a definitive diagnostic differentiation is challenging. Comprehensive evaluations, including history taking, physical examination, audiological and electrophysiological tests, and blood tests, are required for accurate diagnosis. As high-resolution temporal bone CT demonstrates the most consistent and distinctive findings among all reported cases, it is considered the gold standard for diagnosing CCD. CT findings include the absence of a bony wall separating the cochlea from the contiguous carotid canal [4]. MRI and magnetic resonance angiography can be helpful in identifying other diseases that may cause pulsatile tinnitus.
From a surgical perspective, CCD has several important implications. Recognition of CCD prior to otologic procedures, particularly cochlear implantation and stapes surgery, is critical [3,6,7]. During cochlear implantation, an unrecognized CCD significantly increases the risk of misplacing the cochlear electrode array into the carotid canal and damaging the ICA [3]. This can result in a life-threatening hemorrhage. Inadvertent introduction of a cochlear electrode array into the carotid canal during cochlear implantation has previously been reported, even in the absence of a CCD [17]. There are some strategies during electrode array insertion to avoid this catastrophic event. First, the electrode array should advance smoothly along the curvature of the basal turn without any atypical resistance. Second, the use of lubricants such as hyaluronic acid can be helpful for the insertion of the electrode array along the curvature of the cochlea. Third, a perimodiolar-type electrode array may be helpful because its precurved design allows it to hug the modiolus and reduces the risk of inadvertent insertion into the carotid canal.
In terms of stapes surgery, two cases of simultaneous otosclerosis and CCD were reported [6,7]. Both cases showed CHL and Carhart’s notch, suggesting otosclerosis [6,7]. Stapedotomy was unsuccessful in closing the air–bone gap in both cases [6,7]. Subsequent retrospective reassessment of temporal bone CT scans revealed CCD at the apical turn of the cochlea [6,7]. Physicians should consider CCD in the differential diagnosis when evaluating patients with pulsatile tinnitus or atypical otologic symptoms to avoid unnecessary surgical intervention.
Regarding treatment, conservative management could be favorable when symptoms are tolerable [2]. No reports have suggested direct repair of dehiscence because of the high risk of carotid and cochlear damage, which could lead to fatal hemorrhage and deafness [11]. Most reported cases have been managed conservatively with close observation. Medical factors that can generate hyperdynamic circulation, such as anemia, hyperthyroidism, or pregnancy, should be managed appropriately [1]. Obese patients should be evaluated for comorbidities such as HTN, diabetes, and obstructive sleep apnea, and receive proper management and treatment [1,12]. Weight reduction, antihypertensive drugs, blood sugar control, and continuous positive airway pressure are appropriate for each case [12]. Patients should be advised to avoid vigorous physical activity, loud noises, sudden pressure changes, and head trauma. A patient presenting with fluctuating otologic symptoms was successfully treated with a low-salt diet, diuretics, and middle ear pressure control [5]. Some authors suggest that chemical or surgical labyrinthectomy may be considered by destroying the inner ear function of the affected side [2,3]. If the symptoms, especially pulsatile tinnitus and vertigo, are debilitating, and if inner ear function is already impaired, labyrinthectomy could be considered as a treatment option [12]. However, none of the patients underwent labyrinthectomy due to potential complications such as deafness and vestibulopathy. A single case described vestibular neurectomy via the retrosigmoid approach as a hearing-preserving treatment option for eliminating vertigo in a CCD patient presenting with Tullio phenomenon [11]. However, management of pulsatile tinnitus was not described in this patient. In our patient, the only symptom (episodic pulsatile tinnitus) was tolerable and was not associated with HL or vestibular dysfunction. Considering the severity of the symptom and the potential risks and complications of surgical intervention, conservative management was performed. This aligns with prior reports suggesting that conservative management is mostly indicated unless the symptoms are intractable [2].
The natural course of CCD remains unclear, as no reports have documented its prognosis. Disease progression was not reported in most cases. Modugno suggested that bilateral dehiscence may support the hypothesis of a developmental abnormality, as has already been proposed for SCDS [4]. This hypothesis is supported by a case in which temporal bone CT scans obtained 12 years apart showed no interval change in the bony defect [3]. Although dehiscence itself appears stable, chronic transmission of arterial pressure into the perilymph may contribute to progressive inner ear dysfunction in some patients. In our case, the patient experienced episodic pulsatile tinnitus for over 3 years, with a recent increase in its occurrence. As no additional symptoms emerged, it is unlikely that the progression of inner ear injury accounted for the recent worsening of pulsatile tinnitus. Instead, the frequent symptom occurrence may have been caused by a recent increase in physical activity following military enlistment.
Annual follow-ups would be reasonable, consistent with prior reports showing stable symptoms over months to years [3,5]. Additional intervention should be considered if new otologic symptoms arise or if existing symptoms significantly worsen the patient’s quality of life.
To the best of our knowledge, this is the first documented case of CCD in Korea, highlighting the importance of awareness regarding this condition. Because its clinical and audiological manifestations often mimic other otologic and skull base pathologies, definitive differential diagnosis can be challenging. A thorough investigation, including precise evaluation using high-resolution temporal bone CT, is necessary in patients with pulsatile tinnitus. This could be more important preoperatively to avoid fatal complications or unnecessary surgical interventions. This study was approved by the Institutional Review Board (IRB) of The Armed Forces Medical Command (IRB No. AFMC 2025-10-011).
Notes
Acknowledgments
None
Author Contribution
Conceptualization: Ki Hun Jo, Joo Yeon Kim. Data curation: Ki Hun Jo. Investigation: Ki Hun Jo, Joo Yeon Kim, Jae Hwan Kwon. Methodology: Ki Hun Jo. Project administration: Ki Hun Jo, Jae Hwan Kwon. Resources: Ki Hun Jo. Software: Ki Hun Jo, Jong Hyun Choi. Supervision: JY Kim, Jae Hwan Kwon. Validation: Joo Yeon Kim, Jae Hwan Kwon. Visualization: Ki Hun Jo, Jong Hyun Choi. Writing—original draft: Ki Hun Jo. Writing—review & editing: Ki Hun Jo, Jae Hwan Kwon.