Repetitive Transcranial Magnetic Stimulation Protocol for Tinnitus Patients
이명 환자 치료를 위한 경두개 자기 자극 프로토콜
Article information
Abstract
Tinnitus, defined as the perception of sound without an external source, can be particularly distressing when persistent. Among various treatment approaches, repetitive transcranial magnetic stimulation (rTMS) has gained attention as a non-invasive method for modulating maladaptive neuroplastic changes, specifically the hyperactivity and aberrant neural synchrony observed in the auditory cortex (AC) of patients with chronic tinnitus. This report outlines a practical rTMS protocol currently used in clinical settings for tinnitus management. The standard protocol consisted of 1 Hz stimulation at 100%-110% of the resting motor threshold (rMT), applied to the left or contralateral AC, delivering 1800-2000 pulses per session across 10 consecutive treatment days. In selected patients, adjunct excitatory stimulation was delivered to the left dorsolateral prefrontal cortex, especially in cases with comorbid affective symptoms. rTMS offers a safe, adaptable therapeutic option, particularly for patients with concurrent mood disturbances.
Introduction
Tinnitus—the perception of sound without an external source—becomes tinnitus disorder when it triggers autonomic, emotional, or functional impairment [1]. The rationale for repetitive transcranial magnetic stimulation (rTMS) stems from evidence that peripheral hearing loss induces maladaptive central plasticity—specifically, hyperactivity and aberrant synchrony in the auditory cortex (AC)—which may underlie chronic tinnitus [2,3]. Early PET studies showed elevated left AC activity even in the absence of external sound, regardless of tinnitus laterality [4]. Directly suppressing this abnormal activity thus became an early therapeutic goal.
Low-frequency (1 Hz) rTMS inhibits cortical excitability through mechanisms resembling long-term depression [5]. Applying such inhibitory stimulation to the hyperactive AC has become a mainstay of neuromodulatory tinnitus treatment [3]. Beyond the AC, the dorsolateral prefrontal cortex (DLPFC) and anterior cingulate cortex (ACC) are also common targets because tinnitus distress correlates strongly with comorbid depression and anxiety [2,5]. Here we describe our current rTMS protocol for tinnitus.
Methods
Patient selection and informed consent
Written informed consent is obtained before treatment (Fig. 1). General eligibility criteria are as follows.

Representative images of the written informed consent form. A: Page 1, consent statement and study title. B: Page 2, description of risks and benefits. C: Page 3, signature and date.
Inclusion criteria
• Adults ≥18 years with subjective tinnitus
• Tinnitus Handicap Inventory (THI) score ≥38 (at least moderate distress)
Exclusion criteria
• Personal or family history of epilepsy or seizures
• Intracranial metallic implants (e.g., pacemaker, cochlear implant, aneurysm clip)
• Severe traumatic brain injury, prior brain surgery, or stroke
• Pregnancy
• Medications affecting cortical excitability (benzodiazepines, anticonvulsants, neuroleptics)
At our institution, we additionally require documented affective comorbidity: Beck Depression Inventory ≥16, Beck Anxiety Inventory ≥22, or Brief Encounter Psychosocial Instrument-Korean version ≥1.8.
Target localization
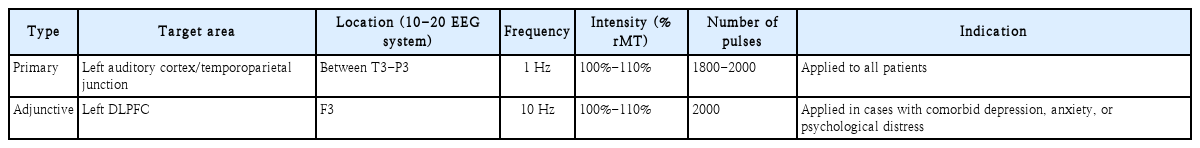
The 10-20 EEG system offers a practical method to approximate the AC, temporoparietal junction (TPJ), and DLPFC (Fig. 2). Electrodes are positioned at 10%, 20%, 20%, 20%, 20%, and 10% intervals along the nasion-inion midline and the interaural line. Common targets are listed in Table 1; in practice, “AC” and “TPJ” refer to the same region. Although MRI-guided neuronavigation theoretically improves targeting precision, clinical outcomes do not differ significantly from those achieved with the 10-20 system [6], likely because the standard figure-of-eight coil stimulates a ~2-cm radius to a depth of 1.5-2.5 cm.
Scalp electrode positions based on the international 10–20 electroencephalography system. Red-circled areas indicate representative sites used in the repetitive transcranial magnetic stimulation protocol. The primary stimulation target for tinnitus is the left temporoparietal junction, typically located between T3 and P3 or T3 and C3/T5. The left dorsolateral prefrontal cortex, marked at F3, is targeted as an adjunct site in cases of comorbid emotional distress. Orientation is based on the midline landmarks (Nasion to Inion and Cz).
Determination of resting motor threshold
The resting motor threshold (rMT) is the lowest TMS intensity that elicits a motor-evoked potential (MEP) ≥50 μV in the thenar muscle or abductor pollicis brevis on ≥50% of trials (e.g., ≥5 of 10) while the muscle is fully relaxed [7]. Accurate rMT calibration ensures both safety and efficacy by individualizing stimulation intensity [8]. Inaccurate measurement risks underdosing (ineffective treatment) or overdosing (increased seizure risk).
Ideally, MEPs are measured with electromyography (EMG). When EMG is unavailable—as in most otolaryngology clinics—visual observation of thumb abduction suffices (Supplementary Video 1). In this setting, rMT is the lowest intensity producing visible abductor pollicis brevis contraction on ≥5 of 10 trials. Though less precise, this method is clinically reliable; reconfirmation in subsequent sessions is advisable. Under adequate lighting, the patient’s hand should rest comfortably to allow clear visualization of thumb movement. All rMT values herein are expressed as a percentage of maximum stimulator output based on visual observation. Average rMT is roughly 50%-60% in women and 55%-65% in men [9,10].
Hotspot-finding procedure
1) Position the coil over the primary motor cortex (-5 cm lateral to the vertex).
2) Begin at subthreshold intensity (e.g., 30% of maximum output).
3) Increase intensity in 5% increments until consistent MEPs appear.
4) Move the coil slightly to identify the scalp site yielding the largest, most consistent MEP at the lowest intensity (the “hotspot”).
5) Fine-tune by reducing output by 1% steps until MEP occurs in <5 of 10 trials, then increase by 1% to confirm ≥5 of 10.
Stimulation parameters
The neurobiological effect depends on frequency: 1 Hz is inhibitory; ≥5 Hz is excitatory. Inhibitory protocols typically use 100%-120% rMT [11]. For example, if rMT is 53%, stimulation at 110% rMT corresponds to 58% (53%×1.1).
Our standard tinnitus protocol delivers 1 Hz at 100%-110% rMT to the left or contralateral AC/TPJ, 1800-2000 pulses per session, over 10 consecutive weekdays (Fig. 3). When adjunctive DLPFC stimulation is indicated, we apply 10-25 Hz at 80%-110% rMT to the left DLPFC (Fig. 4) [11]. For major depressive disorder alone, the standard regimen is 10 Hz to the left DLPFC at 100%-120% rMT, 1600 pulses, over 10-30 sessions; this can be adapted for tinnitus patients with comorbid depression [11].

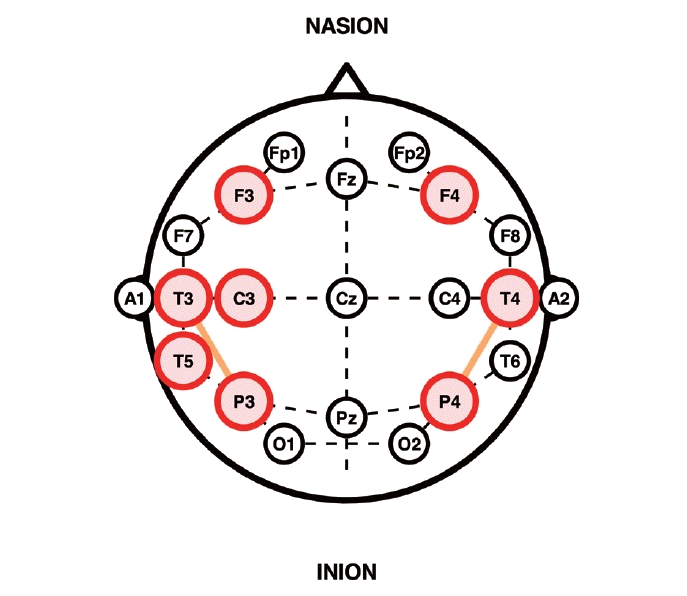
Left temporal cortex coil positioning for 1-Hz repetitive transcranial magnetic stimulation in tinnitus. The coil center is placed over the left auditory cortex/temporoparietal junction target. The coil handle is rotated 45° posterolaterally from the midline so the induced current is approximately perpendicular to the cortical gyrus.
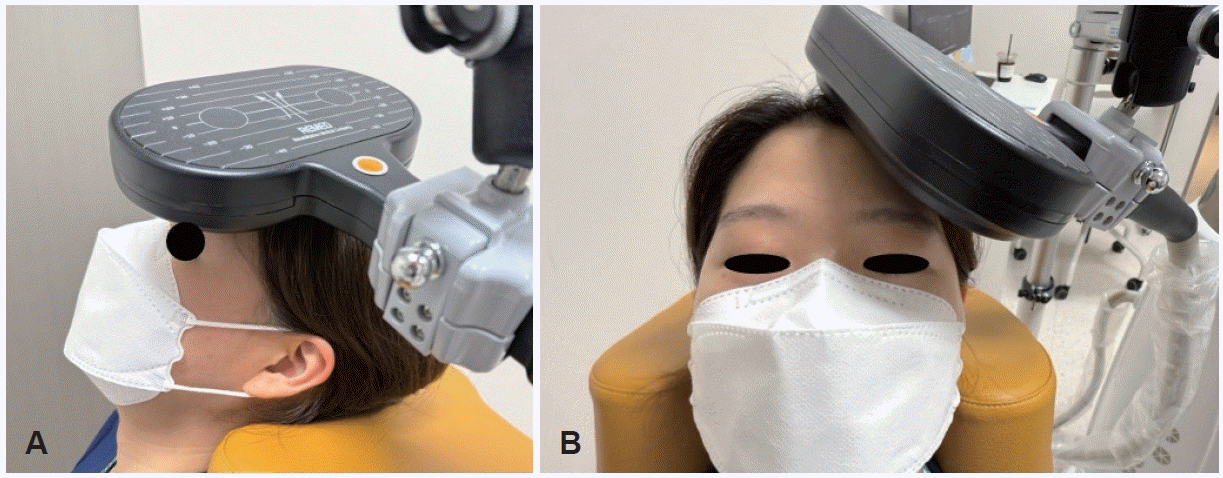
Left dorsolateral prefrontal cortex (DLPFC; F3) coil positioning for high-frequency repetitive transcranial magnetic stimulation. A: Lateral view of placement over F3 based 10–20 electroencephalography system targeting the left DLPFC. B: Frontal/top views of placement over F3. The coil handle is rotated 45° posterolaterally, aligning the induced current near-perpendicular to the cortical gyrus; maintain full coil–scalp contact.
These parameters align with international guidelines. Lefaucheur, et al. [11] assigned level C evidence (possible efficacy) to low-frequency rTMS of the left or contralateral AC for chronic tinnitus and level A evidence (definite efficacy) to high-frequency rTMS of the left DLPFC for depression. Accordingly, we use low-frequency AC stimulation as the primary intervention and add high-frequency DLPFC stimulation in patients with emotional distress or to enhance early adherence. We stimulate DLPFC first—a practical rather than guideline-driven choice based on our clinical experience that it eases anxiety and improves acceptance.
Coil type and orientation
We use a figure-of-eight coil for all procedures. Deep targets such as the ACC may require specialized coils (double-cone or H-coil). Handle orientation affects induced-current direction; we tilt the handle ~45° posterolaterally to direct current perpendicular to the target gyrus, maintaining this orientation consistently.
Treatment delivery and adverse-effect management
During and after each session, patients are monitored for headache, scalp discomfort, facial muscle twitching, or transient tinnitus exacerbation. Seizures are rare but represent the most serious risk. Thorough screening for epilepsy history, seizure-threshold-lowering medications, and substance abuse is essential [12]. Staff must observe patients continuously to detect prodromal signs. If a seizure occurs, stop stimulation immediately, place the patient in the recovery position, protect the head, clear the surroundings, and avoid restraining movements. Most rTMS-induced seizures are self-limiting; benzodiazepines (lorazepam, diazepam, midazolam) should be available, and emergency medical services should be called if needed [13]. A basic emergency kit is mandatory; advanced resuscitation equipment is not routinely required.
Results
As of 2025, we offer rTMS to tinnitus patients who meet the above criteria and desire neuromodulation (Table 2). Between September 2024 and June 2025, 21 patients (8 men, 13 women) completed the protocol. Mean THI decreased from 61.2±20.6 to 28.3±19.3 (p<0.001, paired t-test). Eight patients (38.1%) achieved clinically meaningful improvement (≥20-point THI reduction). One patient experienced tinnitus worsening and discontinued after two sessions. Mild adverse events—scalp pain and headache—occurred in some cases; no serious adverse reactions were observed.
Discussion
Research on rTMS for tinnitus has burgeoned over the past two decades, yet marked heterogeneity persists across studies. Folmer, et al. [14] reported a 56% response rate with effects lasting 26 weeks, whereas a similarly designed multicenter trial by Landgrebe, et al. [15] found no significant difference between active and sham stimulation. Such discrepancies highlight the limitations of a one-size-fits-all approach and have prompted criticism not only of parameter selection but also of oversimplified pathophysiological models.
Initial single-target protocols focused on the AC or TPJ. As evidence emerged linking tinnitus to dysfunctional non-auditory networks—those governing emotion, attention, and memory—the field shifted toward a distributed-network model. This paradigm change spurred multi-site and network-based stimulation strategies.
A prime example is sequential stimulation of the left DLPFC followed by the AC. The DLPFC is an established target in depression; delivering excitatory high-frequency stimulation (10-20 Hz) first may prime emotional regulation, after which inhibitory 1 Hz stimulation to the AC suppresses hyperactivity [11]. That said, some patients respond adequately to AC stimulation alone, and rTMS effects do not always summate—DLPFC stimulation is not mandatory for every patient.
Recently, personalized approaches have gained traction. Brief test sessions using various frequency-location combinations (e.g., 1, 10, or 20 Hz; left or right TPJ) can identify responders via subjective symptom changes (visual analog scale) or EEG markers such as increased alpha and decreased gamma power [16]. Treatment then proceeds with the effective protocol. However, the proportion of patients showing a clear initial response remains low, limiting broader applicability [16].
Theta-burst stimulation (TBS) offers another promising avenue, delivering potent stimulation in shorter sessions. Intermittent TBS is excitatory, and continuous TBS is inhibitory; both exploit theta-rhythm-aligned high-frequency bursts and are under investigation as more efficient protocols [17].
In summary, rTMS for tinnitus has evolved from simple cortical inhibition to a customizable neuromodulatory tool, with ongoing efforts to optimize individualized protocols. Until more effective interventions emerge, rTMS remains a convenient and safe option—particularly for tinnitus patients with comorbid depression.
Supplementary Materials
Korean translation of this article is available with the Online-only Data Supplement at https://doi.org/10.3342/kjorl-hns.2025.00402.
The Supplement is available with this article at https://doi.org/10.3342/kjorl-hns.2025.00402.
Visual estimation of the resting motor threshold (RMT) and identification of the motor hotspot for the right abductor pollicis brevis (APB) in the absence of EMG. With the participant fully relaxed and the hand supported, single-pulse transcranial magnetic stimulation was delivered using a figure-of-eight coil over the left primary motor cortex. The coil was held tangentially with the handle pointing posterolaterally up to 45°. The coil position was adjusted in 0.5-1 cm steps to identify the site that elicited the most consistent visible right thumb abduction - the visual motor hotspot. RMT was defined as the minimal stimulator output producing a visible APB twitch in ≥5 of 10 trials at rest.
Notes
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2024-00352526).