 |
 |
AbstractBackground and ObjectivesFlaps are commonly employed for reconstruction, and many studies have sought to improve flap survival. In this study, we determined whether sildenafil and epinephrine synergistically improve the survival rate of transverse rectus abdominis musculocutaneous (TRAM) flaps.
Materials and MethodTwenty-four Sprague-Dawley rats were divided into four groups of six rats each: a control group, an epinephrine group, a sildenafil group, and an epinephrine-plus-sildenafil group. TRAM flaps were elevated on the upper abdomen.
ResultsThe average flap survival area and number of blood vessels differed significantly between the control and experimental groups; however, the various experimental groups did not differ significantly. The level of vascular epithelial growth factor was higher in the experimental groups than in the control group.
IntroductionHead-and neck surgeons use local, regional, and free flaps to reconstruct defects after extensive cancer resection. Flap necrosis is a major concern. Partial or complete necrosis attributable to flap pedicle issues may trigger infection, delay wound-healing, expose the surgical site, and increase the risk of additional surgery if tissue is exposed to saliva. Flap survival requires a continuous blood supply. Thus, flaps with well-defined vascular anatomies are optimal; surgical delay and administration of various medications immediately after surgery may be appropriate. Flap color and capillary refill time indicate the initial flap condition [1]. Flap blood circulation may be enhanced by vascular dilation and reperfusion, and new blood vessels that develop after surgery. Chemical delay induced by epinephrine is both less invasive than surgical delay and has a more rapid effect. Sildenafil was developed to treat erectile dysfunction, but increases nitric oxide (NO) levels in all tissues, triggering vasodilation. Sildenafil has thus been used recently to reduce ischemia and increase flap survival [2,3]. Sildenafil may also activate the sympathetic neuronal system [4]. It is clear that surgical delay increases flap survival. The utility of vasodilators such as prostaglandin E1 has repeatedly been validated [5]. Here, we explored whether injection of epinephrine into flaps induced vasoconstriction or chemical delay. We aimed to determine whether oral sildenafil citrate activated the sympathetic nervous system and/or promoted vasodilation and angiogenesis. We also examined the combined effects of both drugs on flap survival.
Materials and MethodsThe experiment used 24 Sprague-Dawley rats weighing between 350 to 400 grams, and they were kept under consistent feeding and living conditions. After a one-week adaptation period, in the second week, Group 1 received no treatment, Group 2 received a direct injection of epinephrine (1: 50000) into the transverse rectus abdominis muscle (2 mL), Group 3 received sildenafil (100 mg/tablet) in powdered form, dissolved in 10 mL of normal saline to create a 10 mg/mL solution. This solution was administered orally through a flexible gastric tube at a dose of 0.6-0.8 mL based on each rat’s weight (20 mg/kg) for 7 days. Group 4 received both epinephrine intramuscular injection and sildenafil oral administration. After the drug administration in the second week, each group underwent a transverse rectus abdominis myocutaneous flap procedure in the third week. The results of the flap procedure were analyzed in the fourth week.
Transverse rectus abdominis musculocutaneous (TRAM) flapThe upper border of the flap is positioned approximately 1 cm below the xiphoid process, and the lateral border is aligned with the extension of the anterior axillary line. The lower border of the flap is designed as a rectangular shape (5.0×3.0 cm) based on a parallel line passing through both anterior superior iliac spines. Following the marked flap outline, an incision is made through the skin and subcutaneous tissue (panniculus carnosus) along the flap’s axis. The flap is then elevated from the lateral to medial side above the transverse abdominis fascia, and the lower right rectus abdominis is preserved to serve as the flap pedicle. After elevating the flap and positioning it appropriately, the remaining muscle layers are sutured together using #3-0 vicryl (Ethicon, Bridgewater, New Jersey and Cincinnati, OH, USA) sutures. Subsequently, #4-0 prolene (Ethicon) sutures are used to close the skin (Fig. 1).
Evaluation of flap survivalSeven days after flap creation, digital photographs of the flaps were taken for visual observation. Each photograph was analyzed independently by two different observers using the ImageJ program from the National Institute of Health. The analysis was conducted in a blind test, where the observers were unaware of which group each flap belonged to. In this study, the total area of each flap and the area of the necrotic part of the flap were measured to calculate the ratio. The ratio was expressed as a percentage, representing the extent of necrosis in each group. The necrotic areas were identified as regions of the flap that appeared blackish, dry, and hardened, and did not bleed upon being punctured with a needle. These areas were classified as necrotic. On the other hand, areas that maintained the same appearance as the surrounding skin were considered viable and classified as surviving tissue. By comparing the percentage of necrotic area in each group, differences in flap survival between the experimental groups were assessed.
The Institutional Animal Care and Use Committee (IACUC) of our hospital approved this study (EPSMH2020210101FA).
Histologic assessmentAfter the morphological confirmation, the rats were sacrificed, and the skin flaps were excised. The excised flaps were fixed in a 10% formalin solution for two days. Following formalin fixation, tissue sections were prepared and subjected to hematoxylin and eosin (H&E) staining. The stained samples were observed under an optical microscope at ×40 magnification, focusing on the subcutaneous muscle layer (panniculus carnosus). Subsequently, photographs of each tissue sample were taken at ×100 magnification under an optical microscope, specifically from the dermal layer. The number of blood vessels per unit area was then measured to evaluate vascularity in each tissue sample.
Molecular biological analysisThe tissue, which was stored at -70°C, was placed in 0.5 mL of cell lysis buffer containing proteinase inhibitors (1 μg leupeptin, 1 μg pepstatin A, 1 μg aprotin, and 1 mM PMSF). The cell lysis buffer was composed of 1% Triton X-100, 1% cholic acid, 50 mM NaCl, and 20 mM Tris-HCl, with a pH of 7.4. The tissue was then homogenized using an Ultra-Turrax T25 tissue homogenizer (IKA, Staufen im Breisgau, Germany) for 1 minute with three rounds of homogenization. All tissue processing was performed at 4°C. The solution containing disrupted tissue and cells was subjected to centrifugation (15000 rpm, 15°C, 30 minutes) to separate the nuclear fraction, intact cells, and tissue components, resulting in the precipitation of these fractions and obtaining the supernatant. The cellular and tissue components were then separated, and the protein amount was measured before being analyzed through electrophoresis. Protein quantification was performed using the Bradford method with serum albumin as the standard sample. The protein samples were loaded at a quantity of 25 μg per well onto a 10% polyacrylamide gel. Electrophoresis was conducted at 70 mA for 1 hour. The proteins from the gel were transferred to an Immobilon membrane (Milipore, Bedford, MA, USA). After the transfer, the nitrocellulose membrane was treated with blocking buffer containing 10 mM Tris HCl, 0.15 M NaCl, 0.1% sodium azide, and 5% non-fat dry milk for 1 hour. After the primary antibody reaction using anti-vascular endothelial growth factor (VEGF) mouse monoclonal antibody (1:200) from Santa Cruz (USA) at 4°C overnight, the membrane was further incubated with a horseradish peroxidase-conjugated secondary antibody. The detection of VEGF band was achieved by exposing the membrane to ECL Plus kit (Amersham Life Sciences, Amersham, Buckinghamshire, UK) and visualizing it on a film. Additionally, the optical density of the VEGF band in the Western blot was quantified for further analysis.
Statistical analysisStatistical analysis of the data, including the measurement of the necrotic flap area (%) for the entire group and between each group, was conducted using the SPSS Ver. 21 program (IBM Corp., Armonk, NY, USA). The Kruskal-Wallis test and Mann-Whitney test were used for verification, with a confidence interval of 95% and a significance level set at p<0.05.
ResultsGross findingsThe average flap survival area rates were 66.5% in Group 1 (control), 83.8% in Group 2 (epinephrine), 82.1% in Group 3 (sildenafil citrate), and 84.1% in Group 4 (epinephrine+sildenafil citrate). There was a significant difference between the control group and the experimental groups (p=0.003). However, there was no significant difference among the experimental groups (p=0.567) (Table 1, Figs. 2, 3).
Histologic findingsIn both the experimental and control groups, acute inflammatory findings, characterized by infiltration of mononuclear cells and neutrophils, were observed in the flap necrotic areas. Additionally, in the boundary regions between flap survival and necrosis, granulation tissue and neovascularization were observed. The number of blood vessels per unit area in each group was as follows: Group 1 (control) 13.17±3.312, Group 2 (epinephrine) 22.33±2.733, Group 3 (sildenafil citrate) 24.17±3.189, and Group 4 (epinephrine+sildenafil citrate) 27.83±3.488. The experimental groups showed a significant increase in the number of blood vessels compared to the control group (p=0.001). However, there was no significant difference in the comparison among the experimental groups (p=0.067) (Fig. 4).
Molecular biological findings (VEGF protein expression-optical density)When the value of Group 1 was considered as 1, Group 2 showed 3.19 times higher, Group 3 showed 2.89 times higher, and Group 4 showed 3.11 times higher protein expression compared to the control group. In all cases, protein expression increased compared to the control group. All experimental groups exhibited an increase in protein expression compared to the control group, but the sample size was small, making it difficult to assess statistical significance (Fig. 5).
DiscussionAxial and random pattern flaps are susceptible to hypoxia, hypercarbia, and acidosis attributable to reduced microvasculature flow, in turn triggering metabolic disorders and flap necrosis. To enhance flap survival, it is essential to optimize within-flap blood circulation and thus allow the flap to resist necrosis. Various methods have been used to promote angiogenesis and increase the blood supply, but partial/total flap necrosis after surgery remains distressingly common [6].
Here, we raised transverse rectus abdominis musculocutaneous (TRAM) flaps in Sprague-Dawley rats, as previously reported. In earlier work, the necrotic flap areas of controls that received no pretreatment were 45%-55%. However, our flap survival area was larger. Our flaps were 3×5 cm in area, in contrast to the larger flaps of earlier works (3×4 to 6×10 cm), which were associated with more boundary necrosis [2,7-9]. The drugs (epinephrine and sildenafil citrate) were applied by referring to the existing literature, which experimented with different concentrations and came up with significant conclusions [3,7,10].
We focused on how neovascularization influenced flap survival. Samples were subjected to H&E staining and the number of blood vessels per unit area was counted under an optical microscope. The number increased significantly in all three experimental groups compared to the control group. However, in the experimental groups, it was difficult to distinguish between the expansion of existing blood vessels and the opening of previously closed capillary networks. VEGF ameliorates angiogenesis induced by hypoxia. All experimental groups exhibited increased VEGF expression, as revealed by Western blotting, but the small sample numbers did not allow assessment of the statistical significance of group differences.
Delayed treatment improves flap survival, although why this improves blood circulation remains unclear. The flap gradually becomes resistant to hypoxia, and ischemia stimulates the vascular system. Capillary networks that were closed during the resting state open; as treatment is further delayed, venous outflow becomes obstructed, increasing blood flow in the superficial capillaries and thus improving blood circulation [11]. In other words, when a flap is raised in a step-by-step manner, ischemia is prevented. During the final stage of each step, materials secreted by the sympathetic nervous system are metabolized. Blood vessel constriction is relieved, increasing the likelihood of flap survival and reducing metabolic demand; this improves flap survival.
After creating and elevating the flap, the effect of delayed treatment is apparent after 8-10 days. At this point, the flap is less likely to become ischemic. At 10-14 days after flap elevation, reinnervation occurs, triggering closure of the venous outflow and thus permitting successful flap transfer. The beneficial effects of delayed treatment are thought to persist for 2-3 weeks in humans [6,8].
Epinephrine injection into the flap is a form of drug-induced delayed treatment. Although epinephrine can be applied in several ways, systemic administration of epinephrine is thought to be of limited clinical use in some patients, especially as it may affect the cardiovascular system, and intraperitoneal injection was ruled out to eliminate confounding variables due to the possibility of using ketamine anesthesia. Epinephrine acts on the alpha-adrenergic receptors of skin blood vessels to trigger sustained vasoconstriction, and thus ischemia, which is associated with induction of hypoxia-inducible factor 1 alpha (HIF-1α). In turn, HIF-1α stimulates VEGF expression, which promotes neovascularization. Flap circulation increases, improving the chance of survival [12]. According to Karacaoglu, et al. [7], direct subcutaneous administration of epinephrine is also a good alternative to surgical delay.
Other drug-based methods are used to increase flap survival. An increase in the deformability of red blood cells reduces the viscosity of capillary flow, thus enhancing the flap blood supply. Local or systemic endothelial cell growth factor administration after surgery increases flap survival. Thus, various drug-based interventions enhance flap survival by improving blood circulation and promoting angiogenesis [13-15].
Sildenafil citrate is a phosphodiesterase-5 inhibitor that activates the NO guanylate cyclase pathway to inhibit phosphodiesterase-5 (PDE-5) enzyme activity. This enzyme converts cyclic guanosine monophosphate (cGMP) to GMP and thus suppresses NO depletion caused by reactive oxygen species. Thus, body levels of NO and cGMP remain high for extended periods. High NO levels inhibit platelet aggregation and vasodilation, but promote neovascularization, thus increasing flap survival. cGMP stimulates HIF-1α-mediated VEGF transactivation, in turn inducing angiogenesis that further enhances flap survival [16]. Tsai, et al. [3] found that intraperitoneal sildenafil citrate at 10 mg/kg/day increased flap survival. Sarifakioglu, et al. [10] used oral sildenafil citrate at 3, 10, and 20 mg/kg to this end. Hart, et al. [17] reported the short-and long-term effects of sildenafil citrate on flap survival. In summary, sildenafil citrate enhances flap survival and is a valuable treatment option. Single-dose sildenafil citrate delivered orally or intravenously does not significantly change the arterial pressure or heart rate, but oral administration increases the activity of the sympathetic nervous system and plasma catecholamine levels [4,18,19]. Sildenafil citrate can cross the blood-brain barrier and inhibit the PDE-5 activity of brain blood vessels, thus directly impacting the central nervous system and increasing the activity of the sympathetic nervous system [20].
We explored the effects of combined epinephrine and sildenafil citrate on flap survival through both sympathetic nervous system activation and vasodilation. Compared to the control group, the experimental groups had significant increases in both average flap survival area and the number of blood vessels; both drugs were effective. However, there was no significant difference between the group that received only one drug and the group given both drugs. Based on the results of VEGF protein expression, it is thought that the alpha-receptor action of epinephrine may cause continuous vasoconstriction, and the resulting ischemia may induce neovascularization, but the vasodilatory function of sildenafil citrate by NO does not cause ischemia, and only VEGF-induced angiogenesis remains, so it may not be possible to show a significant difference from the sole effect. No prior study found that drug interaction affected flap survival. The effects of nicotine and sildenafil citrate on flap survival have been compared, and both drugs induced ischemia. Nicotine compromised vascular relaxation and reduced NO levels, whereas epinephrine did not [21]. The synergistic effects of the two drugs are not clear, and clinical evidence for their combined use seems to be lacking, but each drug has already been proven in several studies. In the future, we plan to test other drugs known to increase flap survival (dexamethasone, botulinum toxin A etc), either alone or in combination, to increase flap survival and reduce side effects.
The limitations of this study include the fact that the digital photographs did not clearly distinguish necrotic from surviving flap areas, although two blinded observers analyzed all images. Thus, there may have been observer bias. Additionally, during flap surgery and harvesting, it was impossible to completely blind all researchers to the experimental group allocations. This creates a risk of bias in terms of the surgical technique chosen.
In conclusion, we placed right inferior pedicle TRAM flaps in rats; both epinephrine and sildenafil citrate increased flap survival and the number of blood vessels per unit area, and VEGF enhanced neovascularization. However, no combined effect of epinephrine and sildenafil citrate was apparent; further research is required.
ACKNOWLEDGMENTSThis work was supported by grants from the alumni associated with the Department of Otolaryngology Head and Neck Surgery, The Catholic University of Korea (No. 5-2022-B0001-00024).
This research was funded by The Catholic University of Korea, Eunpyeong St. Mary’s Hospital Research Institute of Medical Science.
NotesAuthor contributions Conceptualization: Dong-Hyun Lee, Jun-Ook Park. Data curation: Dong-Hyun Lee, Jiyoung Yeo, Jun-Ook Park. Formal analysis: Dong-Hyun Lee, Jiyoung Yeo. Funding acquisition: Jun-Ook Park. Investigation: Dong-Hyun Lee, J Yeo, Soomin Park, Sung-Ho Cho, Subin Kim. Methodology: Dong-Hyun Lee, Jun-Ook Park. Project administration: Dong-Hyun Lee, Jiyoung Yeo. Resources: Jiyoung Yeo, Soomin Park, Sung-Ho Cho, Subin Kim. Software: Jiyoung Yeo. Supervision: Ji-Sun Kim, Byung Guk Kim, Ki-Hong Chang, Jun-Ook Park. Validation: Dong-Hyun Lee, Ji-Sun Kim, Ki-Hong Chang. Writing—original draft: Dong-Hyun Lee, Jiyoung Yeo. Writing—review & editing: Dong-Hyun Lee, Soomin Park, Subin Kim, Ji-Sun Kim, Byung Guk Kim, Jun-Ook Park. Fig. 1.Transverse rectus abdominis musculocutaneous (TRAM) flap model marking of the TRAM flap in the rat abdomen (A), flap after elevation (B), closure of the abdominal wall defect (C) view of the flap after the operation (D). 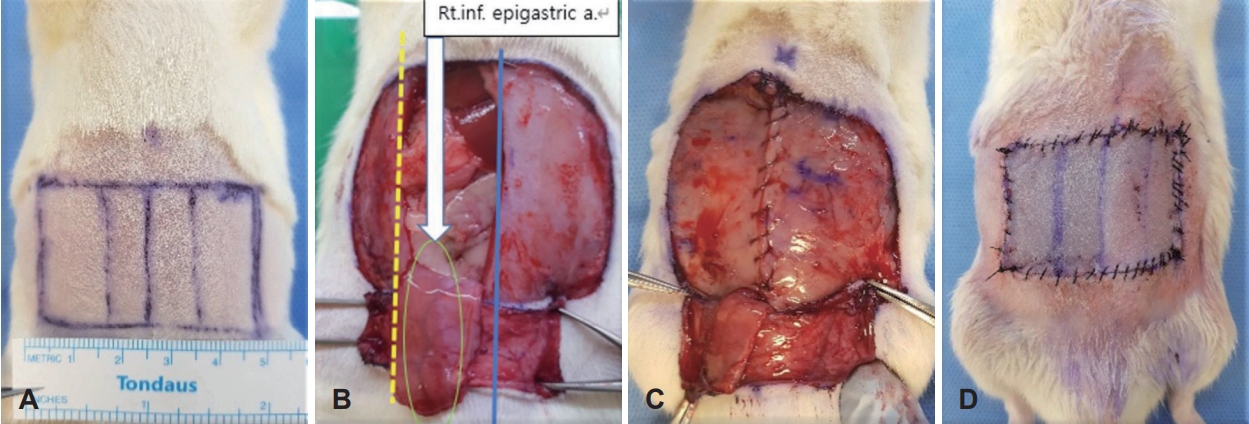
Fig. 2.Gross and histologic findings. Measuring the survival area ratio by ImageJ software (A) hematoxylin and eosin stains of tissue sample (blue arrow: vessel) (B). 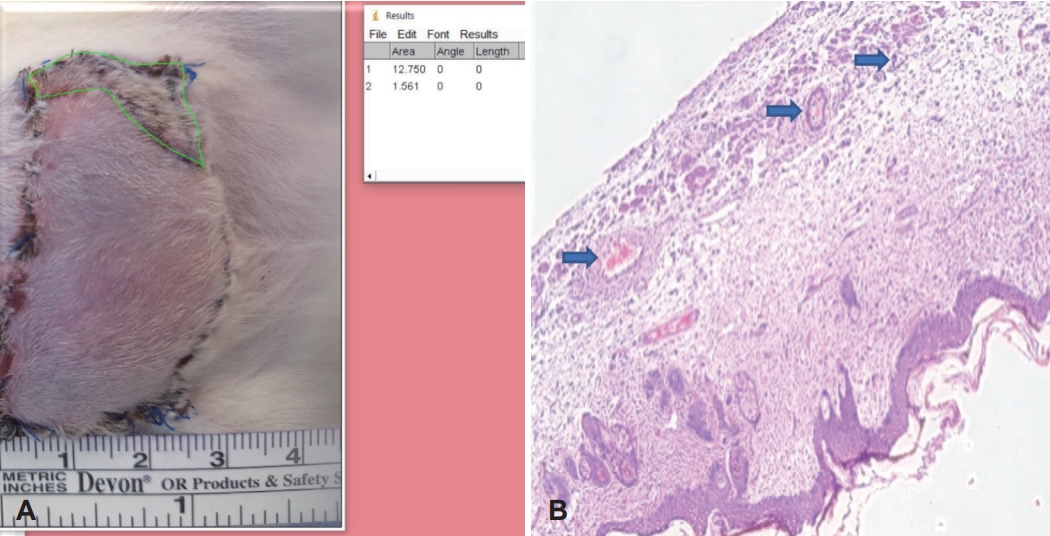
Fig. 3.The mean percentage of the flap survival of the experimental groups (Group 1: control, Group 2: epinephrine, Group 3: sildenafil, Group 4: epinephrine+sildenafil). 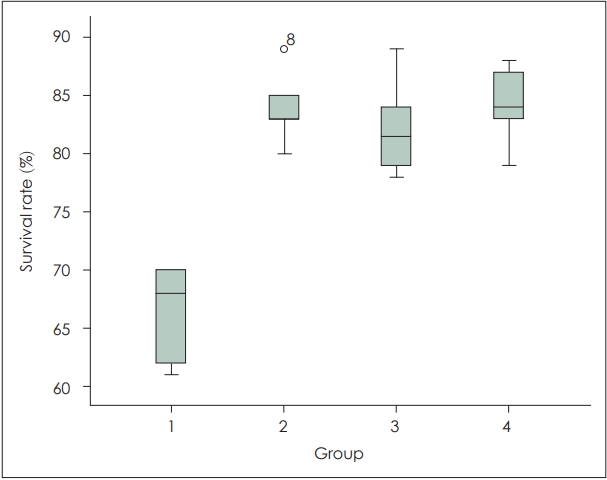
Fig. 4.Comparison of the flap vessels number in upper dermis of each experimental group (Group 1: control, Group 2: epinephrine, Group 3: sildenafil, Group 4: epinephrine+sildenafil). 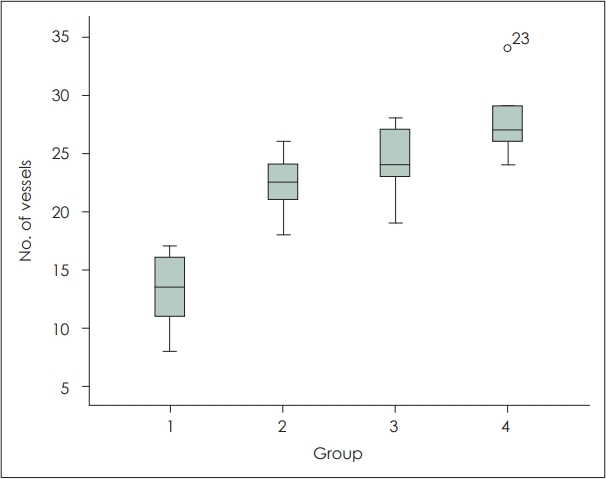
Fig. 5.Comparison of the vascular endothelial growth factor (VEGF) protein expression using western blot method (Group 1: control, Group 2: epinephrine, Group 3: sildenafil, Group 4: epinephrine+ sildenafil). GAPDH, glyceraldehyde-3-phosphate dehydrogenase. 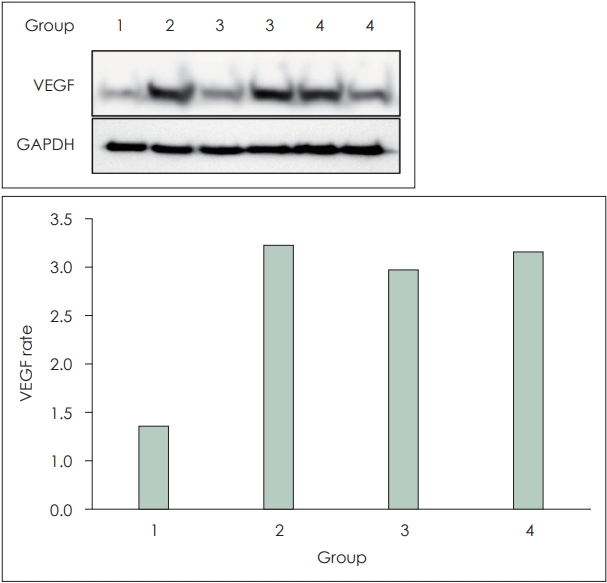
Table 1.Survival rate and number of number of vessels in the four groups REFERENCES1. Mahieu R, Colletti G, Bonomo P, Parrinello G, Iavarone A, Dolivet G, et al. Head and neck reconstruction with pedicled flaps in the free flap era. Acta Otorhinolaryngol Ital 2016;36(6):459-68.
2. Ozgentaş HE, Shenaq S, Spira M. Study of the delay phenomenon in the rat TRAM flap model. Plast Reconstr Surg 1994;94(7):1018-24, discussion 1025-6.
3. Tsai JW, Ayubi FS, Hart KL, Baur DA, Parham MA, Moon JK, et al. Evaluation of the effect of sildenafil and vascular endothelium growth factor combination treatment on skin flap survival in rats. Aesthetic Plast Surg 2008;32(4):624-31.
4. Phillips BG, Kato M, Pesek CA, Winnicki M, Narkiewicz K, Davison D, et al. Sympathetic activation by sildenafil. Circulation 2000;102(25):3068-73.
5. Asai S, Fukuta K, Torii S. Topical administration of prostaglandin E1 with iontophoresis for skin flap viability. Ann Plast Surg 1997;38(5):514-7.
6. Pearl RM. A unifying theory of the delay phenomenon--recovery from the hyperadrenergic state. Ann Plast Surg 1981;7(2):102-12.
7. Karacaoglu E, Yuksel F, Turan SO, Zienowicz RJ. Chemical delay: An alternative to surgical delay experimental study. Ann Plast Surg 2002;49(1):73-80.
8. Cinpolat A, Bektas G, Coskunfirat N, Rizvanovic Z, Coskunfirat OK. Comparing various surgical delay methods with ischemic preconditioning in the rat TRAM flap model. J Reconstr Microsurg 2014;30(5):335-42.
9. Costa W, Silva AL, Costa GR, Vidigal PV, Pereira FH. Analysis of effect isoxsuprine hydrochloride and nicotine in the transverse rectus abdominis myocutaneous flap (TRAM) in rats. Acta Cir Bras 2015;30(7):497-502.
10. Sarifakioglu N, Gokrem S, Ates L, Akbuga UB, Aslan G. The influence of sildenafil on random skin flap survival in rats: An experimental study. Br J Plast Surg 2004;57(8):769-72.
11. Hallock GG, Rice DC. Evidence for the efficacy of TRAM flap delay in a rat model. Plast Reconstr Surg 1995;96(6):1351-7.
12. Krammer CW, Ibrahim RM, Hansen TG, Sørensen JA. The effects of epinephrine and dobutamine on skin flap viability in rats: A randomized double-blind placebo-controlled study. J Plast Reconstr Aesthet Surg 2015;68(1):113-9.
13. Wu ZJ, Ibrahim MM, Sergesketter AR, Schweller RM, Phillips BT, Klitzman B. The influence of topical vasodilator-induced pharmacologic delay on cutaneous flap viability and vascular remodeling. Plast Reconstr Surg 2022;149(3):629-37.
14. Pickens JP, Rodman SM, Wetmore SJ. The effects of extended perioperative pentoxifylline on random skin flap survival. Am J Otolaryngol 1994;15(5):358-69.
15. Pu LL, Ahmed S, Thomson JG, Reid MA, Madsen JA, Restifo RJ. Endothelial cell growth factor enhances musculocutaneous flap survival through the process of neovascularization. Ann Plast Surg 1999;42(3):306-12.
16. Cakmak E, Karasoy Yesilada A, Sevim KZ, Sumer O, Tatlidede HS, Sakiz D. Effect of sildenafil citrate on secondary healing in full thickness skin defects in experiment. Bratisl Lek Listy 2014;115(5):267-71.
17. Hart K, Baur D, Hodam J, Lesoon-Wood L, Parham M, Keith K, et al. Short- and long-term effects of sildenafil on skin flap survival in rats. Laryngoscope 2006;116(4):522-8.
18. Gillies HC, Roblin D, Jackson G. Coronary and systemic hemodynamic effects of sildenafil citrate: From basic science to clinical studies in patients with cardiovascular disease. Int J Cardiol 2002;86(2-3):131-41.
19. Raja SG, Nayak SH. Sildenafil: Emerging cardiovascular indications. Ann Thorac Surg 2004;78(4):1496-506.
|
|
|||||||||||||||||||||||||||||||||||||||

 |
 |