 |
 |
AbstractPapillary thyroid carcinoma (PTC) is a lymphotropic cancer in which cervical nodal metastasis may precede the detection of the primary tumor. Incidental cervical masses are increasingly identified with the wider use of ultrasonography in local clinics. Although PTC is often first recognized from a cervical mass, it is generally assumed to originate in the thyroid. Very rarely, metastatic PTC presents without an identifiable thyroid primary, and only a few such cases have been reported worldwide. We recently managed an incidentally detected metastatic PTC without a demonstrable primary tumor. A 50-year-old male presented with a cervical mass detected on ultrasonography. This case report outlines diagnostic considerations and management without postoperative radioactive iodine treatment for metastatic PTC of unknown primary with cervical lymphatic involvement.
IntroductionPapillary thyroid carcinoma (PTC) is the most common thyroid malignancy, accounting for approximately 80% of cases [1]. It is lymphotropic: clinically evident cervical nodal metastasis occurs in about 20%-50% of patients [2]. Historically, many patients presented with an asymptomatic cervical mass; more recently, incidental masses have been detected during ultrasonography performed for health checkups. Even when PTC is first suspected from a cervical mass rather than a thyroid lesion, the primary is typically in the thyroid gland. Exceptionally, metastatic PTC presents without a detectable thyroid primary, with only a few cases reported worldwide. We describe a rare case of incidentally discovered metastatic PTC with no identifiable primary tumor in the thyroid.
CaseA 50-year-old male was referred to our outpatient clinic after incidentally detected cervical masses during an ultrasound-guided procedure for neck pain at a local clinic 3 weeks ago. Local ultrasonography revealed heterogeneous masses of varying sizes in the right neck. He was asymptomatic, had no underlying disease, and no palpable mass was detected on physical examination. On the day of presentation, repeat ultrasonography demonstrated a 2.04×1.11 cm oval-shaped, well-defined, heterogeneously hypoechoic mass with low vascularity and loss of the fatty hilum at right level III (Fig. 1). The sonographic appearance did not specifically suggest metastatic PTC; rather, a neurogenic tumor was favored because of the mass morphology and perivascular location. No suspicious lesions were identified in the thyroid parenchyma or salivary glands. Because pathologic confirmation was required to guide management, fine-needle aspiration of the mass was performed simultaneously and revealed metastatic PTC; nuclear enlargement, multiple nuclear pseudoinclusions, and grooves were noted with positive immunohistochemical staining for thyroid transcription factor-1 (Fig. 2).
The biopsy report was explained to the patient and after counseling, the patient elected surgery to remove a presumed occult thyroid primary and any additional nodal disease. Contrast-enhanced neck CT was subsequently obtained to evaluate the thyroid gland and additional cervical metastases before surgery and no other imagaing study was performed. No definite lesion was identified in both thyroid lobe, and only a single enlarged right level III lymph nodes were observed (Fig. 1). Total thyroidectomy with right selective neck dissection (levels II-IV) and central compartment node dissection was performed in accordance with 2015 American Thyroid Association (ATA) guidelines to remove hidden malignancy in thyroid gland, to enable postoperative serum thyroglobulin (Tg) monitoring for recurrence detection and to allow radioactive iodine (RAI) ablation if needed [3]. A firm 3.0×2.0 cm mass was noted at right level III, and a firm 0.5 cm nodule at the superior thyroid isthmus was suspicious for micro-PTC. Both recurrent laryngeal nerves were identified and preserved. Pathologic examination showed no abnormality in either thyroid lobe, and the palpable isthmic nodule represented nodular hyperplasia (Fig. 3). Total 11 lymph nodes of 28 were found metastatic PTC and single node with extranodal extension (ENE) at right level II was identified with 18 mm of largest dimension; metastatic involvement included 5 level VI nodes of 9 without ENE, 5 right level II nodes of 7 with ENE, and 1 right level III node of 8 without ENE; no level IV nodes were involved (0/4). Postoperatively, both the resected lymph nodes and the isthmic nodular hyperplasia were tested for BRAF mutation, and all results were negative.
Postoperative positron emission tomography (PET)-CT, thyroid scintigraphy with technetium uptake (Fig. 4), and Tg measurement were performed to assess for residual thyroid tissue or ectopic thyroid gland. PET-CT detected no additional metastatic lesions; the technetium thyroid scan showed no visible residual thyroid tissue; and the Tg level was 0.27 ng/mL, below our laboratory’s lower limit of normal. No further treatment was indicated after multidisciplinary consultation. The patient recovered without complications and remains under routine follow-up without recurrence.
DiscussionCervical metastasis from PTC without an identifiable intrathyroidal primary is rare but well documented. A lateral neck mass should prompt consideration of nodal metastasis; among such lesions, thyroid malignancy is the second most common, particularly in individuals younger than 40 years without other risk factors [4].
Several mechanisms may explain cervical metastasis in PTC when the primary tumor is not identified: 1) a microcarcinoma smaller than the histologic section could be missed on microscopy; 2) the nodal disease may arise from an ectopic thyroid carcinoma or from ectopic thyroid tissue in other organs; and 3) the primary tumor may have regressed over time.
The total thyroidectomy specimen was processed according to standard grossing protocols. After fixation in 10% neutral buffered formalin for 24-48 hours, measurements were recorded and resection margins were inked. Serial sectioning was initially performed at 4 mm intervals in a sagittal or transverse plane. As no definite primary lesion was identified, additional thin sectioning less than 2 mm intervals was carried out for thorough sampling of the entire thyroid parenchyma to detect an occult primary lesion, yet no primary tumor was found. In a study of 82 patients initially believed to have solitary PTC on preoperative ultrasonography, entire thyroids were sectioned horizontally at 2 mm, embedded in paraffin, and cut into 4 mm slides from each block. This detailed mapping revealed 66 cases of occult PTC, corresponding to an incidence of 15.8% in Korea, with most (97.0%) measuring 3 mm or less [5]. Thus, micro-PTC can be missed when the primary focus is smaller than the slide thickness, suggesting that thinner-than-routine sections may help detect otherwise occult malignancy.
Approximately 1%-3% of ectopic thyroids occur in the lateral neck, with an estimated incidence of about 1 in 300000 individuals. Carcinomas arising in ectopic thyroid tissue are exceedingly rare (<1% of all thyroid cancers), and PTC is the most common subtype [6]. Accordingly, ectopic thyroid carcinoma should be excluded pathologically in cases of cervical PTC, despite its rarity. In our patient, postoperative pathology confirmed that the cervical mass was a nodal metastasis, and postoperative PET-CT and thyroid scan showed no residual thyroid tissue. Additionally, about 10% of ectopic thyroid tissue has been reported in the esophagus, heart, lung, aorta, mediastinum, duodenum, or gallbladder [7]. Because there was no thyroid primary and the cervical lesion represented metastasis, metastasis from ectopic thyroid tissue also had to be excluded. When an intrathyroidal primary is not identified, PET-CT may be recommended to evaluate for metastasis from ectopic thyroid carcinoma, although coverage limitations within some insurance systems can restrict its use.
Spontaneous regression of the primary tumor could also have occurred within the thyroid. Under active surveillance for PTC, 6.7% of patients experienced a tumor size decrease exceeding 50% of the original, and tumor volume could not be calculated in 2.4% [8]. Activation of the patient’s immune system may contribute to regression by inducing fibrosis around the primary site with inflammatory cell infiltration [9]. However, neither fibrosis nor inflammatory infiltration was observed in our specimen, underscoring the need for further study of silent tumor regression.
The patient is currently undergoing routine follow-up without additional treatment. According to the 2015 ATA guidelines for the management of thyroid nodules and cancer, RAI ablation may be considered for intermediate-risk patients and should be individualized. The strength of the recommendation varies according to clinical details; discussions should incorporate each patient’s clinicopathologic features, risk of recurrence, multidisciplinary clinical judgment and Tg levels. ATA guideline also suggests serial Tg level monitoring serves as a reliable surrogate marker for recurrence detection, allowing earlier identification [3]. In a retrospective cohort study, there was no difference in the 5-year recurrence-free survival among intermediate-risk patients who did or did not receive radioiodine therapy when postoperative unstimulated serum Tg levels were <1 ng/mL [10]. Additionally, the absence of BRAF mutation, which is associated with aggressive clinicopathologic features and increased recurrence risk in PTC, also supports active surveillance without additional therapy in this patient [3]. Moreover, RAI ablation has not been shown to reduce locoregional recurrence risk even in intermediate-risk PTC patients with aggressive features such as BRAF positivity, larger tumors, multifocality, extrathyroidal extension, or regional lymph node metastasis [11]. Recent studies additionally show conflicting evidence on adjuvant RAI benefits in intermediate risk PTC and notably, no disease-specific survival benefit was observed with RAI in N1b PTC patients <55 years, even with intermediate-risk features [12]. Given the patient’s low postoperative serum Tg level, negativity for the BRAF mutation, age, absence of residual metastatic thyroid tissue on PET-CT, and regular multidisciplinary follow-up in both our clinic and endocrinology department, observation without RAI ablation remains reasonable [10].
Metastatic PTC without a detectable primary is exceedingly rare worldwide, though several cases have been reported. We reviewed past 15-year case reports of ATA intermediate-risk metastatic PTC without primary tumors (Table 1) [13-15]. All cases received postoperative RAI ablation per institutional protocols; only one mentioned Tg levels, used solely to confirm no recurrence rather than guide RAI decisions. Unlike prior reports, this is the first to show recurrence-free outcomes despite omitting RAI, supporting de-escalation even in rare cases. However, as a case report with inherently limited statistical power, further studies are warranted to establish clinical stability and long-term efficacy.
NotesAuthor Contribution Conceptualization: Yong-Dae Kim, Si-Youn Song. Data curation: Gi Moon Yoo. Formal analysis: Gi Moon Yoo, Jaebeom Park. Investigation: Yong-Dae Kim. Methodology: Gi Moon Yoo, Jaebeom Park, Si-Youn Song. Project administration: Gi Moon Yoo, Jaebeom Park. Supervision: Yong-Dae Kim, Si-Youn Song. Validation: Si-Youn Song. Visualization: Yong-Dae Kim, Si-Youn Song. Writing—original draft: Gi Moon You. Writing—review & editing: Gi Moon Yoo. Fig. 1.Preoperative ultrasonography and CT findings. A: An oval-shaped, well-defined, heterogeneously hypoechoic mass measuring 2.04×1.11 cm with low vascularity and loss of the hilum is present at right cervical level III. B-D: Axial (B and C) and coronal (D) contrastenhanced CT images show an approximately 14×15×21 mm, well-marginated, heterogeneously enhancing solid mass-like lesion at right level III (red arrows) and no definite space-occupying lesion in either thyroid lobe. 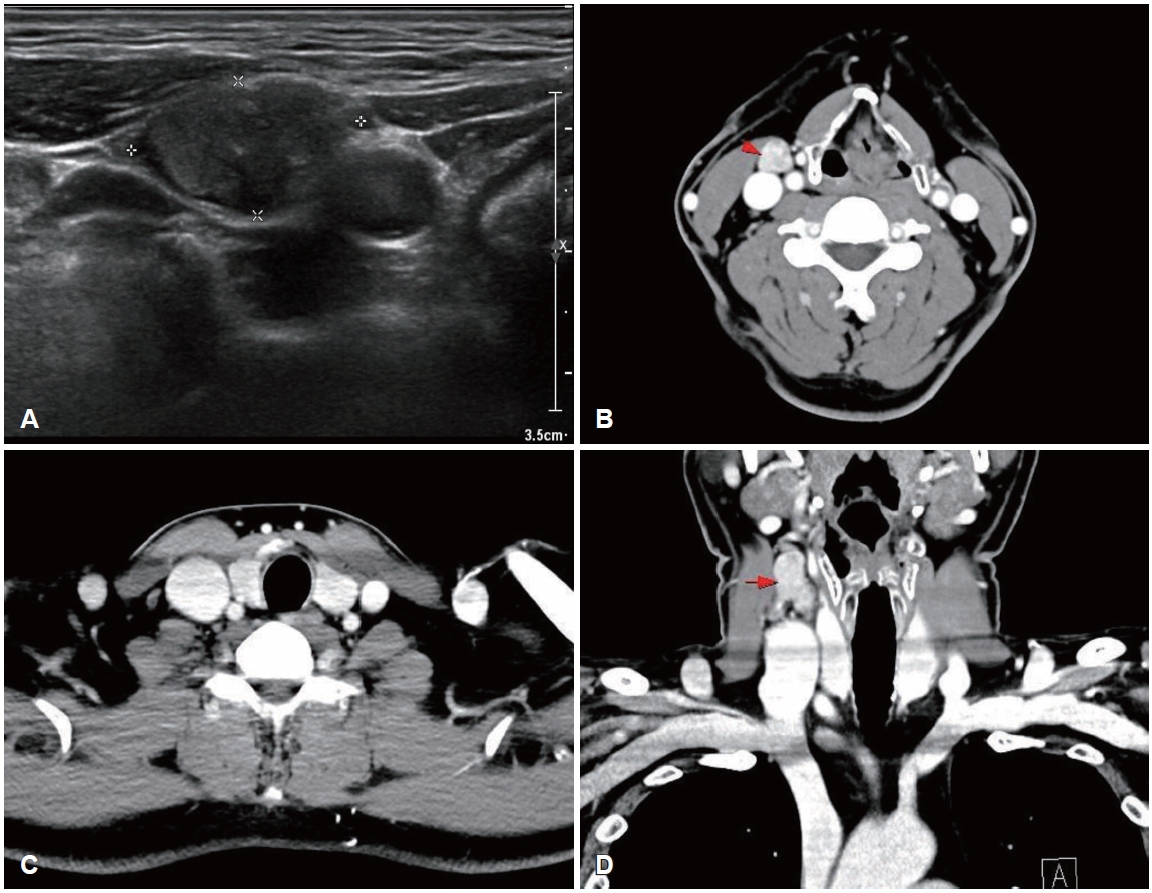
Fig. 2.Histologic features of the right level III mass from fine-needle aspiration. A: Histopathology shows nuclear enlargement, multiple nuclear pseudoinclusions, and grooves, consistent with metastatic papillary thyroid carcinoma (hematoxylin-eosin, 400×). B: Immunohistochemical staining for thyroid transcription factor-1 was positive in the tumor cells (hematoxylin-eosin, 200×). 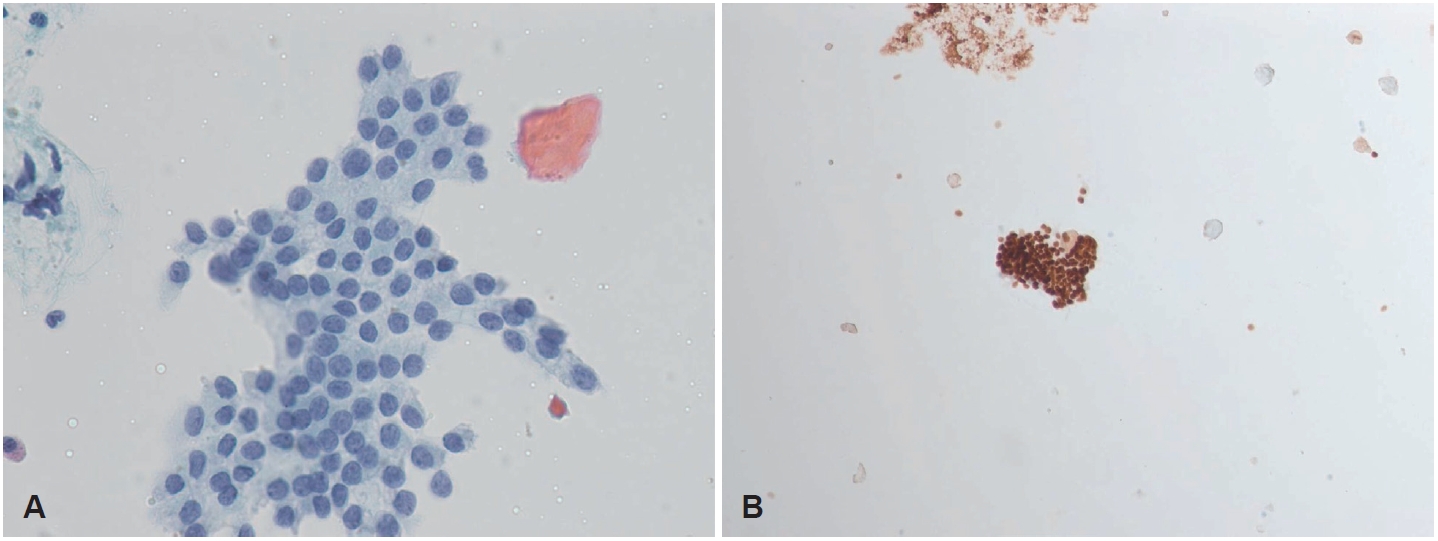
Fig. 3.Histologic features of the thyroid gland. Histopathology showed 0.2×0.2 cm nodular hyperplasia at thyroid isthmus without features of papillary thyroid carcinoma (hematoxylin-eosin, 40×). 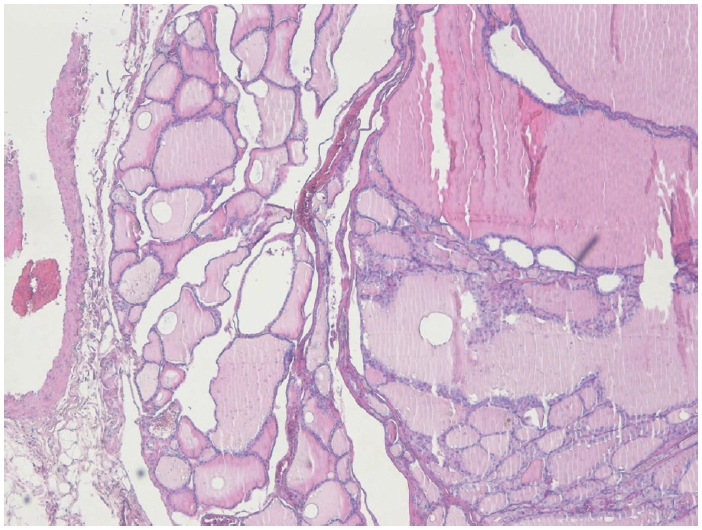
Fig. 4.Postoperative positron emission tomography-CT and thyroid scan findings. A and B: Uneven 18F-fluorodeoxyglucose uptake in the anterior neck and mild uptake in both lateral necks suggest postoperative changes and reactive lymph nodes. No other hypermetabolic lesions suggest metastasis to other organs. C: Postoperative Tc-99m pertechnetate thyroid scan shows no significant radioactive accumulation. 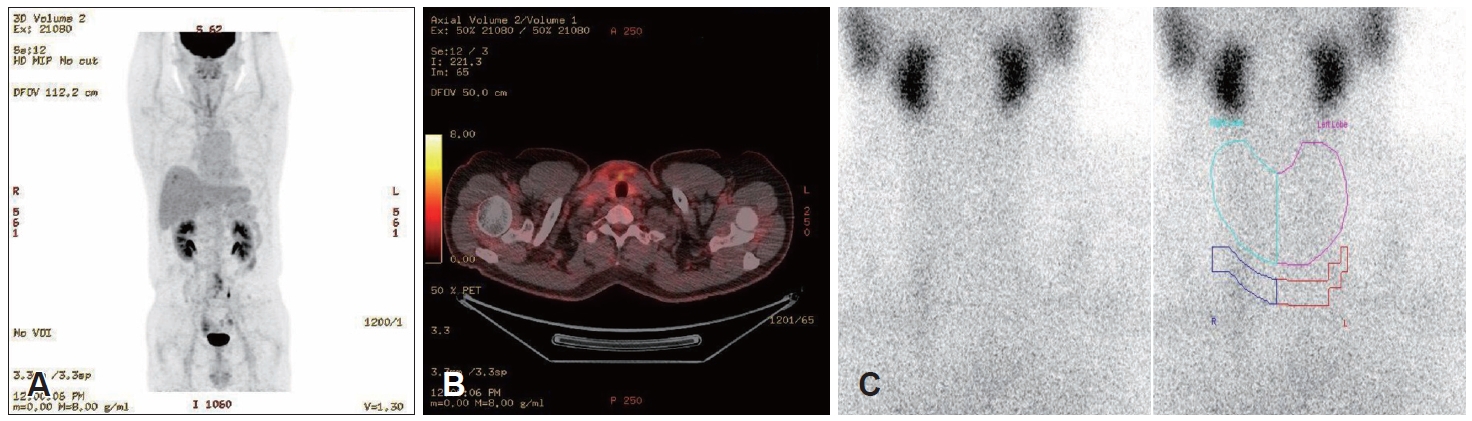
Table 1.Previous reports of ATA intermediate-risk metastatic PTC without primary tumors
Previous case reports of ATA intermediate-risk metastatic PTC without primary tumors in past 15-years. ATA, American Thyroid Association; PTC, papillary thyroid carcinoma; TT, total thyroidectomy; CCND, central compartment node dissection; SND, selective neck disscecion; LND, lateral neck dissection; RAI, radioactive iodine; Tg, thyroglobulin. REFERENCES2. Yan S, Yu J, Zhao W, Wang B, Zhang L. Prophylactic bilateral central neck dissection should be evaluated based on prospective randomized study of 581 PTC patients. BMC Endocr Disord 2022;22(1):5.
3. Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 2016;26(1):1-133.
4. Garrel R, Tripodi C, Cartier C, Makeieff M, Crampette L, Guerrier B. Cervical lymphadenopathies signaling thyroid microcarcinoma. Case study and review of the literature. Eur Ann Otorhinolaryngol Head Neck Dis 2011;128(3):115-9.
5. Park SY, Jung YS, Ryu CH, Lee CY, Lee YJ, Lee EK, et al. Identification of occult tumors by whole-specimen mapping in solitary papillary thyroid carcinoma. Endocr Relat Cancer 2015;22(4):679-86.
6. Agosto-Vargas Y, Gutiérrez M, Martínez JH, Mangual-Garcia M, Palermo C, Vélez-Maymi S, et al. Papillary thyroid carcinoma: ectopic malignancy versus metastatic disease. Case Rep Endocrinol 2017;2017:9707031.
7. Qi Y, Liu J, Liu Y, Shen Z, Hu N. Ectopic papillary thyroid carcinoma mimicking distant metastatic tissue. J Int Med Res 2022;50(9):3000605221121968.
8. Tuttle RM, Fagin JA, Minkowitz G, Wong RJ, Roman B, Patel S, et al. Natural history and tumor volume kinetics of papillary thyroid cancers during active surveillance. JAMA Otolaryngol Head Neck Surg 2017;143(10):1015-20.
9. Jung SH, Park JH, Kim J, Choi N. A case of metastatic papillary thyroid carcinoma in neck lymph nodes without primary tumor in thyroid gland. Ear Nose Throat J 2025;104(2_suppl):340S-5.
10. Ibrahimpasic T, Nixon IJ, Palmer FL, Whitcher MM, Tuttle RM, Shaha A, et al. Undetectable thyroglobulin after total thyroidectomy in patients with low- and intermediate-risk papillary thyroid cancer—is there a need for radioactive iodine therapy? Surgery 2012;152(6):1096-105.
11. Kim SK, Woo JW, Lee JH, Park I, Choe JH, Kim JH, et al. Radioactive iodine ablation may not decrease the risk of recurrence in intermediate-risk papillary thyroid carcinoma. Endocr Relat Cancer 2016;23(5):367-76.
12. Palacardo F, Lee-Saxton YJ, Tumati A, Marshall TE, Greenspun BC, Zarnegar R, et al. The impact of radioactive iodine on disease-specific survival in low-to-intermediate risk N1b papillary thyroid carcinoma. Ann Surg Oncol 2025;32(3):1698-708.
13. Carrillo A, Arbab F, Ramani NS. Metastatic papillary thyroid carcinoma in the lymph nodes without identifiable primary tumor in the thyroid. Cureus 2025;17(5):e84554.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |